Evaluating the role of Salivary α-Amylase and Associated Enzymes in Carbohydrate Digestion and Gastrointestinal Function: A Comprehensive Systematic Review Perspective
Keywords:
Salivary α-Amylase, Disaccharidase, Carbohydrate Metabolism, Gastrointestinal Disorders, Systematic Review, Metabolic HealthAbstract
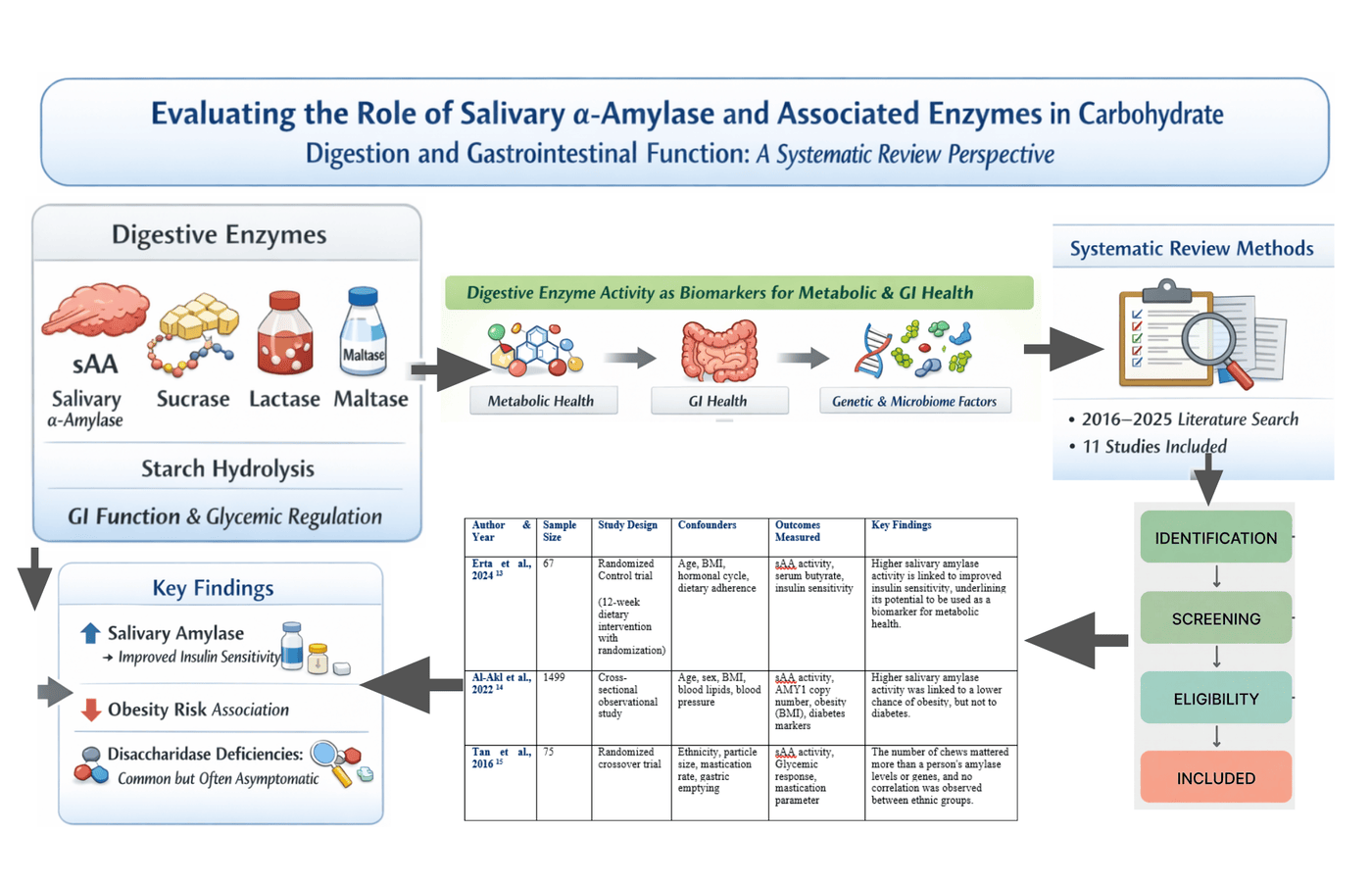
Background: Salivary α-amylase, sucrase, lactase, and maltase enzymes demonstrate an important role in carbohydrate metabolism and gastrointestinal function. Changes in enzymatic activity impact starch digestion, gut fermentation, insulin sensitivity, and digestive health. This systematic review aimed to evaluate how salivary α-amylase, sucrase, lactase, and maltase levels influence metabolic outcomes, carbohydrate digestion, and gastrointestinal functions. Methods: This systematic review followed PRISMA guidelines 2020. Research was conducted in PubMed, Scopus, Web of Science, and Google Scholar from 2016 to 2025. The studies involving salivary α-amylase, sucrase, lactase, and maltase activities in relation to metabolic and gastrointestinal outcomes were included. Non-human research or studies lacking outcome measurements were excluded. The appropriate tools were used for evaluating risk of bias according to the study designs whereas, quality of study was evaluated using GRADE approach. Results: The included eleven studies revealed that higher levels of salivary α-amylase, sucrase, lactase, and maltase were linked to improved gastrointestinal functions, such as greater insulin sensitivity and a lower risk of obesity. The risk of bias of most studies was measured as low to moderate and GRADE rated certainty of evidence as moderate. Conclusion: Salivary α-amylase, sucrase, lactase, and maltase activities might be useful indicators of metabolic health and digestion. Additional research is needed to clarify their use in clinical applications and personalized nutrition.
References
1. Meng J, Sun Y, Wu P, Dong Z, Qin Y, Wang L, et al. Physiologically relevant simulation of carbohydrate digestion: From glycemic index estimation to intestinal cellular responses. Foods. 2025;14(22):3864. https://doi.org/10.3390/foods14223864
2. Lejk A, Myśliwiec K, Jarosz-Chobot P. Effects of different types of meals on postprandial glycaemia in healthy subjects. Pediatr Endocrinol Diabetes Metab. 2024;30(1). https://doi.org/10.5114/pedm.2024.142587
3. Borowitz D. Non-pancreatic digestive enzymes. Biomolecules. 2025;15(9):1259. https://doi.org/10.3390/biom15091259
4. Kim SB, Calmet FH, Garrido J, Garcia-Buitrago MT, Moshiree B. Sucrase-isomaltase deficiency as a potential masquerader in irritable bowel syndrome. Dig Dis Sci. 2020;65(2):534–540. https://doi.org/10.1007/s10620-019-05780-7
5. Fernández-Bañares F. Carbohydrate maldigestion and intolerance. Nutrients. 2022;14(9):1923. https://doi.org/10.3390/nu14091923
6. Willis HJ, Asche SE, McKenzie AL, Adams RN, Roberts CG, Volk BM, et al. Impact of continuous glucose monitoring versus blood glucose monitoring to support a carbohydrate-restricted nutrition intervention in people with type 2 diabetes. Diabetes Technol Ther. 2025;27(5):341–356. https://doi.org/10.1089/dia.2024.0406
7. Qi X, Tester RF. Lactose, maltose, and sucrose in health and disease. Mol Nutr Food Res. 2020;64(8):1901082. https://doi.org/10.1002/mnfr.201901082
8. Singh MP, Agrawal NR, Saurabh S, Krishna E, Singh JM. Exploring therapeutic digestive enzyme landscape in India: current evidence, profit motives, regulations, and future perspectives. Cureus. 2024;16(1). https://doi.org/10.7759/cureus.52891
9. Khalid S, Amjad Y, Zafar L, Khan O, Shahzadi L, Rafa SA. Systematic review: diagnosis, treatment and management strategies of lactose intolerance. Open Access Res J Bio Pharm. 2024;12(1):001–13. https://doi.org/10.53022/oarjbp.2024.12.1.0032
10. Hashash JG, Squire J, Francis FF, Binion DG, Cross RK, Farraye FA. An expert opinion/approach: clinical presentations, diagnostic considerations, and therapeutic options for gastrointestinal manifestations of common variable immune deficiency. Am J Gastroenterol. 2022;117(11):1743–1752. https://doi.org/10.14309/ajg.0000000000002027
11. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71
12. Carra MC, Romandini P, Romandini M. Risk of bias evaluation of cross-sectional studies: adaptation of the Newcastle–Ottawa Scale. J Periodontal Res. 2025;60(2):e13405. https://doi.org/10.1111/jre.13405
13. Erta G, Gersone G, Jurka A, Tretjakovs P. The link between salivary amylase activity, overweight, and glucose homeostasis. Int J Mol Sci. 2024;25(18):9956. https://doi.org/10.3390/ijms25189956
14. Al-Akl N, Thompson RI, Arredouani A. Elevated levels of salivary α-amylase activity in saliva associated with reduced odds of obesity in adult Qatari citizens: a cross-sectional study. PLoS One. 2022;17(3):e0264692. https://doi.org/10.1371/journal.pone.0264692
15. Tan VMH, Ooi DSQ, Kapur J, Wu T, Chan YH, Henry CJ, et al. The role of digestive factors in determining glycemic response in a multiethnic Asian population. Eur J Nutr. 2016;55(4):1573–1581. https://doi.org/10.1007/s00394-015-0976-0
16. Dbar S, Akhmadullina O, Sabelnikova E, Belostotskiy N, Parfenov A, Bykova S, et al. Patients with functional bowel disorder have disaccharidase deficiency: a single-center study from Russia. World J Clin Cases. 2021;9(17):4178. https://doi.org/10.12998/wjcc.v9.i17.4178
17. Deb C, Campion S, Derrick V, Ruiz V, Abomoelak B, Avdella A, et al. Sucrase-isomaltase gene variants in patients with abnormal sucrase activity and functional gastrointestinal disorders. J Pediatr Gastroenterol Nutr. 2021;72(1):29–35. https://doi.org/10.1097/MPG.0000000000002852
18. Dale HF, Hagen M, Bekkelund M, Deb C, Valeur J. Disaccharidase deficiencies and gastrointestinal symptoms in patients referred to gastroscopic examination: a single-center study from Norway. Scand J Gastroenterol. 2024;59(10):1166–1171. https://doi.org/10.1080/00365521.2024.2395848
19. Kemple B, Rao SS. Disaccharidase enzyme deficiency in adult patients with gas and bloating. Clin Transl Gastroenterol. 2025;16(3):e00809. https://doi.org/10.14309/ctg.0000000000000809
20. Viswanathan L, Rao SS, Kennedy K, Sharma A, Yan Y, Jimenez E. Prevalence of disaccharidase deficiency in adults with unexplained gastrointestinal symptoms. J Neurogastroenterol Motil. 2020;26(3):384. https://doi.org/10.5056/jnm19167
21. Colombo JM, Friesen CS, Garg U, Friesen CA, Pablo WS. Relationships between disaccharidase deficiencies, duodenal inflammation and symptom profile in children with abdominal pain. Sci Rep. 2021;11(1):4902. https://doi.org/10.1038/s41598-021-84535-9
22. Erta G, Gersone G, Jurka A, Tretjakovs P. Decoding metabolic connections: the role of salivary amylase activity in modulating visceral fat and triglyceride glucose index. Lipids Health Dis. 2025;24(1):98. https://doi.org/10.1186/s12944-025-02524-7
23. Erta G, Gersone G, Jurka A, Tretjakovs P. Impact of a 12-week dietary intervention on adipose tissue metabolic markers in overweight women of reproductive age. Int J Mol Sci. 2024;25(15):8512. https://doi.org/10.3390/ijms25158512
24. Huang PC, Chen HC, Huang HB, Lin YL, Chang WT, Leung SH, et al. Mediating effects of insulin resistance on lipid metabolism with elevated paraben exposure in the general Taiwan population. Front Public Health. 2025;13:1416264. https://doi.org/10.3389/fpubh.2025.1416264
25. Zahra SA, Butt YN, Nasar S, et al. Food packaging in perspective of microbial activity: a review. J Microbiol Biotechnol Food Sci. 2016;6(2):752–757. https://doi.org/10.15414/jmbfs.2016.6.2.752-757
26. Aldossari NM, El Gabry EE, Gawish GE. Association between salivary amylase enzyme activity and obesity in Saudi Arabia. Medicine (Baltimore). 2019;98(23):e15878. https://doi.org/10.1097/MD.0000000000015878
27. Al-Akl NS, Thompson RI, Arredouani A. Reduced odds of diabetes associated with high plasma salivary α-amylase activity in Qatari women: a cross-sectional study. Sci Rep. 2021;11(1):11495. https://doi.org/10.1038/s41598-021-90977-y
28. Khoso SA, Khan S, Aslam N, Memon S, Muneer G, Ahmed F, et al. Analysis of saliva flow rate and pH from addictive users of cohort of Hyderabad and adjoining area. World J Pharm Res. 2017;6(3):143–156. https://doi.org/10.20959/wjpr20173-7930
29. Cash BD, Patel D, Scarlata K. Demystifying carbohydrate maldigestion: a clinical review. Am J Gastroenterol. 2025;120(4S):1–11. https://doi.org/10.14309/ajg.0000000000003374
30. Majeed M, Majeed S, Nagabhushanam K, Arumugam S, Pande A, Paschapur M, et al. Evaluation of the safety and efficacy of a multienzyme complex in patients with functional dyspepsia: a randomized, double-blind, placebo-controlled study. J Med Food. 2018;21(11):1120–1128. https://doi.org/10.1089/jmf.2017.4172
31. Medernach J, Middleton JP. Malabsorption syndromes and food intolerance. Clin Perinatol. 2022;49(2):537–555. https://doi.org/10.1016/j.clp.2022.02.015
32. Ferreira-Lazarte A, Moreno FJ, Villamiel M. Bringing the digestibility of prebiotics into focus: update of carbohydrate digestion models. Crit Rev Food Sci Nutr. 2021;61(19):3267–3278. https://doi.org/10.1080/10408398.2020.1798344
33. Proença C, Ribeiro D, Freitas M, Fernandes E. Flavonoids as potential agents in the management of type 2 diabetes through the modulation of α-amylase and α-glucosidase activity: a review. Crit Rev Food Sci Nutr. 2022;62(12):3137–3207. https://doi.org/10.1080/10408398.2020.1862755
34. Clemente-Suárez VJ, Mielgo-Ayuso J, Martín-Rodríguez A, Ramos-Campo DJ, Redondo-Flórez L, Tornero-Aguilera JF. The burden of carbohydrates in health and disease. Nutrients. 2022;14(18):3809. https://doi.org/10.3390/nu14183809
35. Pérez-Ros P, Navarro-Flores E, Julián-Rochina I, Martínez-Arnau FM, Cauli O. Changes in salivary amylase and glucose in diabetes: a scoping review. Diagnostics (Basel). 2021;11(3):453. https://doi.org/10.3390/diagnostics11030453
36. Wagan G, Bakhsh L, Mehmood A, Bano S, Memon S, Samo RP. Effects of red meat consumption on histology of coronary arteries in adult albino mice. Ann Punjab Med Coll. 2020;13(4):287–91. https://doi.org/10.29054/apmc/2019.760
37. Qazi N, Memon S, Memon F, Goswami P, Sirhandi BR, Goswami B. Antioxidant and hepato-protective effects of ginger in comparison with atorvastatin in hyperlipidemic albino mice. JMMC. 2024;15(1):21–26. https://doi.org/10.62118/jmmc.v15i1.464
38. Viswanathan L, Rao SS. Intestinal disaccharidase deficiency in adults: evaluation and treatment. Curr Gastroenterol Rep. 2023;25(6):134–139. https://doi.org/10.1007/s11894-023-00870-z
39. Catanzaro R, Sciuto M, Marotta F. Lactose intolerance—old and new knowledge on pathophysiological mechanisms, diagnosis, and treatment. SN Compr Clin Med. 2021;3(2):499–509. https://doi.org/10.1007/s42399-021-00792-9
40. Montoro-Huguet MA, Belloc B, Domínguez-Cajal M. Small and large intestine (I): malabsorption of nutrients. Nutrients. 2021;13(4):1254. https://doi.org/10.3390/nu13041254
41. Lushchak VI. Symphony of digestion: coordinated host–microbiome enzymatic interplay in gut ecosystem. Biomolecules. 2025;15(8):1151. https://doi.org/10.3390/biom15081151
42. Rotaru M, Singeap AM, Ciobica A, Huiban L, Stanciu C, Romila L, et al. Oral health and “modern” digestive diseases: pathophysiologic and etiologic factors. Biomedicines. 2024;12(8):1854. https://doi.org/10.3390/biomedicines12081854
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Ehsan Khan, Sabeeha Saddique, Syeda Maira Gillani, Sitara Ejaz, Sidra Anwar, Uday Shree Akkala (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





