A Comprehensive Meta-Analytical Evidence on IFN-γ and TNF-α as Diagnostic and Treatment Monitoring Biomarkers in Tuberculosis
Keywords:
Tuberculosis, Interferon-gamma, Tumor Necrosis Factor-alpha, Biological Markers, Diagnosis, Treatment, Monitoring, Systematic ReviewAbstract
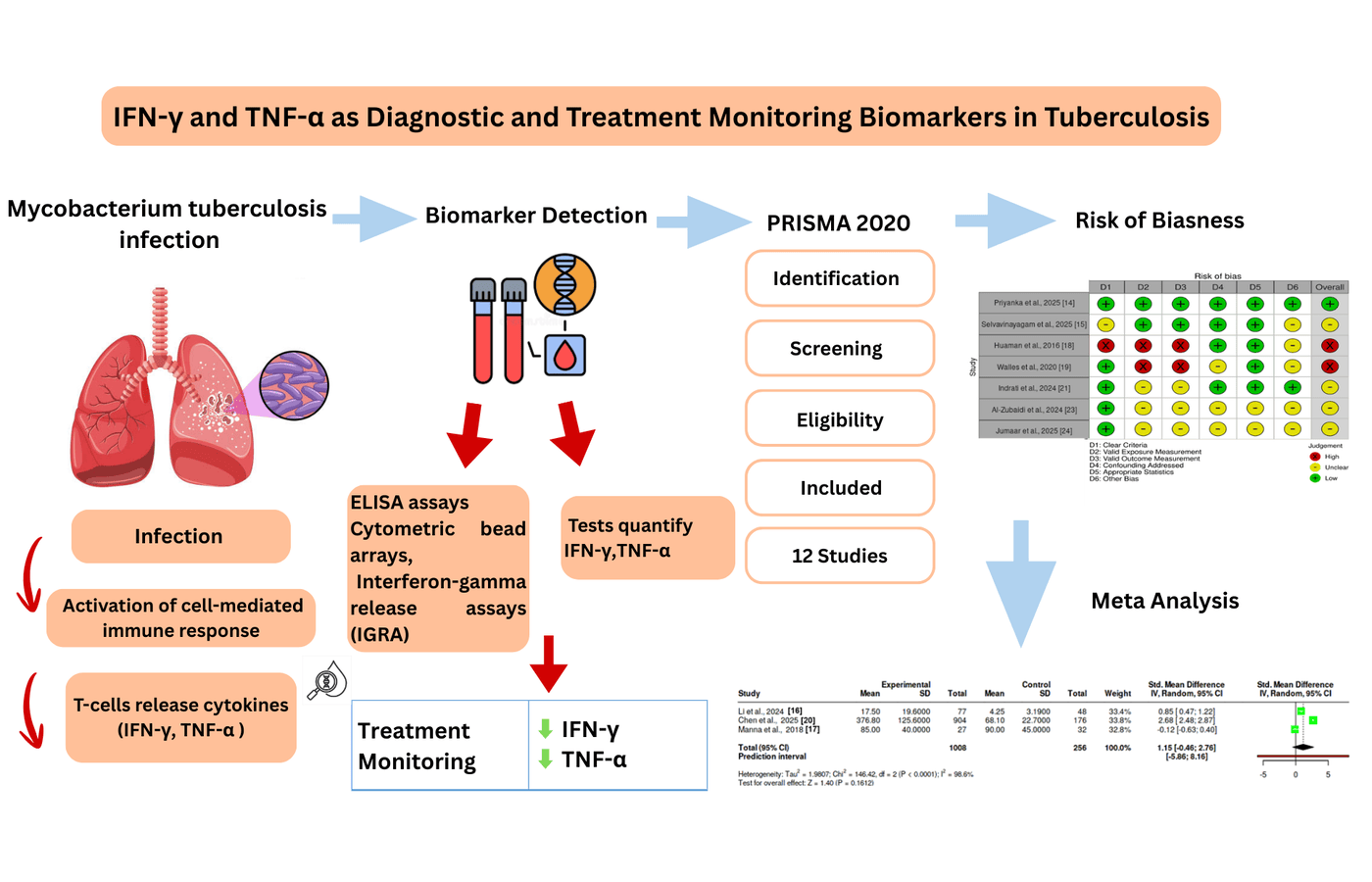
Background: The accurate diagnosis and effective surveillance of tuberculosis (TB) is a global health issue. The responsible cytokines in host immune response against Mycobacterium tuberculosis were interferon-gamma (IFN-γ) and tumor necrosis factor -alpha (TNF-α). This study was aimed at synthesizing the existing evidence on diagnostic accuracy of IFN-γ and TNF-α levels in TB, and to evaluate their role in monitoring treatment response. Methods: This review followed PRISMA 2020 guidelines for its synthesis. Relevant published articles from in PubMed, Scopus, Web of Science, and Google Scholar from 2016 to 2025 were analyzed. The included studies were those that compared circulating IFN-γ and/or TNF-α levels in patients with TB, with or without treatment. Review-based studies, and studies without comparable biomarker data were excluded. For risk of bias assessment, Newcastle-Ottawa Scale was utilized, and the certainty of evidence was measured through GRADE framework. Results: The analysis of 12 included studies demonstrated that high levels of IFN-γ were a uniform biomarker of diagnosis for latent and active TB infection, whereas higher levels of TNF-α level were associated with active pulmonary TB. Longitudinal data showed that reduction in both cytokine levels was associated with successful treatment outcomes, while their persistent elevated levels suggested poor outcomes or relapse. Risk of bias among studies was low to moderate, and certainty of evidence was found to be moderate. Conclusion: IFN-γ was a good diagnostic biomarker throughout the TB spectrum, TNF-α was promising in the detection of active disease and assessment of therapeutic response.
References
1. Hange N, Somagutta MR, Sharma A, Agadi K, Ngaba NN, Paikkattil N, et al. Latent tuberculosis: challenges and opportunities for diagnosis and treatment. Discov Med. 2022;5(1):e27. https://doi.org/10.15190/drep.2022.1
2. Palanivel J, Sounderrajan V, Thangam T, Rao SS, Harshavardhan S, Parthasarathy K. Latent tuberculosis: challenges in diagnosis and treatment, perspectives, and the crucial role of biomarkers. Curr Microbiol. 2023;80(12):392. https://doi.org/10.1007/s00284-023-03491-x
3. Yuk J-M, Kim JK, Kim IS, Jo E-K. TNF in human tuberculosis: a double-edged sword. Immune Netw. 2024;24(1):e4. https://doi.org/10.4110/in.2024.24.e4
4. Shanmuganathan G, Orujyan D, Narinyan W, Poladian N, Dhama S, Parthasarathy A, et al. Role of interferons in Mycobacterium tuberculosis infection. Clin Pract. 2022;12(5):788-96. https://doi.org/10.3390/clinpract12050082
5. Arias AA, Neehus A-L, Ogishi M, Meynier V, Krebs A, Lazarov T, et al. Tuberculosis in otherwise healthy adults with inherited TNF deficiency. Nature. 2024;633(8029):417-25. https://doi.org/10.1038/s41586-024-07866-3
6. Yilmaz E, Pasin Ö, Pasin T. Screening potential of tuberculin skin and interferon gamma release tests for latent tuberculosis prior to anti-tumor necrosis factor (TNF)-α therapy in patients with rheumatoid arthritis and spondyloarthritis. Egypt Rheumatol. 2025;47(1):31-5. https://doi.org/10.1016/j.ejr.2024.11.003
7. Zachariou M, Arandjelović O, Sloan DJ. Automated methods for tuberculosis detection/diagnosis: a literature review. BioMedInformatics. 2023;3(3):724-51. https://doi.org/10.3390/biomedinformatics3030047
8. Kontsevaya I, Cabibbe AM, Cirillo DM, DiNardo AR, Frahm N, Gillespie SH, et al. Update on the diagnosis of tuberculosis. Clin Microbiol Infect. 2024;30(9):1115-22. https://doi.org/10.1016/j.cmi.2023.07.014
9. Datta K, LaRue R, Permpalung N, Das S, Zhang S, Mehta Steinke S, et al. Development of an interferon-gamma release assay (IGRA) to aid diagnosis of histoplasmosis. J Clin Microbiol. 2022;60(10):e01128-22. https://doi.org/10.1128/jcm.01128-22
10. Yang Y, Wang H-J, Hu W-L, Bai G-N, Hua C-Z. Diagnostic value of interferon-gamma release assays for tuberculosis in the immunocompromised population. Diagnostics. 2022;12(2):453. https://doi.org/10.3390/diagnostics12020453
11. Parums DV. Review articles, systematic reviews, meta-analysis, and the updated preferred reporting items for systematic reviews and meta-analyses (PRISMA) 2020 guidelines. Med Sci Monit. 2021;27:e934475-1. https://doi.org/10.12659/MSM.934475
12. Fekete JT, Győrffy B. MetaAnalysisOnline. com: web-based tool for the rapid meta-analysis of clinical and epidemiological studies. J Med Internet Res. 2025;27:e64016. https://preprints.jmir.org/preprint/64016
13. Carra MC, Romandini P, Romandini M. Risk of bias evaluation of cross-sectional studies: Adaptation of the Newcastle–Ottawa Scale. J Periodontal Res. 2025 Apr;60(2):e13405. https://doi.org/10.1111/jre.13405
14. Priyanka, Bhatt P, Bedi AK, Basil MV, Sharma M, Sharma S. Association of latent tuberculosis and Vitamin D levels in young individuals: An exploratory study. Int J Mycobacteriol. 2025;14(1):66-72. https://doi.org/10.4103/ijmy.ijmy_6_25
15. Selvavinayagam ST, Sankar G, Yong YK, Sankar S, Zhang Y, Tan HY, et al. Association of clinical laboratory parameters with latent tuberculosis infection among healthcare workers of primary health centers―A cross-sectional observational study. PLOS Glob Public Health. 2025;5(6):e0004873. https://doi.org/10.1371/journal.pgph.0004873
16. Li X, Zhao W, Han H, Yang Z, Bi F, He Y. Retrospective analysis of the clinical utility of multi-cytokine profiles in smear-negative pulmonary tuberculosis. Saudi Med J. 2024;45(7):658. https://doi.org/10.15537/smj.2024.45.7.20240310
17. Manna MP, Orlando V, Li Donni P, Sireci G, Di Carlo P, Cascio A, et al. Identification of plasma biomarkers for discrimination between tuberculosis infection/disease and pulmonary non tuberculosis disease. PLoS One. 2018;13(3):e0192664. https://doi.org/10.1371/journal.pone.0192664
18. Huaman MA, Deepe Jr GS, Fichtenbaum CJ. Elevated circulating concentrations of interferon-gamma in latent tuberculosis infection. Pathog Immun. 2016;1(2):291. https://doi.org/10.20411/pai.v1i2.149
19. Walles J, Tesfaye F, Jansson M, Balcha TT, Sturegård E, Kefeni M, et al. Tuberculosis infection in women of reproductive age: a cross-sectional study at antenatal care clinics in an Ethiopian city. Clin Infect Dis. 2021;73(2):203-10. https://doi.org/10.1093/cid/ciaa561
20. Chen J, Deng B, Li M, Huang Y, Han J, Li Q, et al. Analysis of the clinical value and influencing factors of combined detection of tuberculosis-specific cytokines IFN-γ and IL-2 in the diagnosis of active tuberculosis in children and adolescents. Eur J Med Res. 2025;30(1):732. https://doi.org/10.1186/s40001-025-02895-8
21. Indrati AR, Sumarpo A, Haryanto J, Rosmiati NMD, Munaya S, Turbawaty DK, et al. Identification of cytokine signatures in HIV‑infected individuals with and without Mycobacterium tuberculosis co‑infection. Biomed Rep. 2024;21(3):131. https://doi.org/10.3892/br.2024.1819
22. Nie W, Wang J, Jing W, Shi W, Wang Q, Huang X, et al. Value of serum cytokine biomarkers TNF-α, IL-4, sIL-2R and IFN-γ for use in monitoring bacterial load and anti-tuberculosis treatment progress. Cytokine: X. 2020;2(2):100028. https://doi.org/10.1016/j.cytox.2020.100028
23. Al-Zubaidi MI, Lafi SA, Abdulateef YM. Cytokine Dysregulation in pulmonary Tuberculosis: The role of TNF-α/IL-10 and TNF-α/TGF-β ratios as severity indicators. Hum Immunol. 2025;86(2):111256. https://doi.org/10.1016/j.humimm.2025.111256
24. Jumaar C, Malefane L, Jacobs S, Sanni O, Louw E, Baines N, et al. Delineating the Significance of Several Inflammatory Markers in a Lung Tuberculosis Cohort During the Active and Post-Tuberculosis Stages of the Disease: An Observational Study in Cape Town, South Africa (2019 to 2024). Infect Dis Rep. 2025;17(3):52. https://doi.org/10.3390/idr17030052
25. Waghmare PJ, Lende T, Goswami K, Gupta A, Gupta A, Gangane N, et al. Immunological host responses as surveillance and prognostic markers in tubercular infections. Int J Mycobacteriol. 2019;8(2):190-5. https://doi.org/10.4103/ijmy.ijmy_48_19
26. Khanna H, Gupta S, Sheikh Y. Cell-Mediated Immune Response Against Mycobacterium tuberculosis and Its Potential Therapeutic Impact. J Interferon Cytokine Res. 2024;44(6):244-59. https://doi.org/10.1089/jir.2024.0030
27. Singha SP, Memon S, Kazi SAF, Nizamani GS. Gamma aminobutyric acid signaling disturbances and altered astrocytic morphology associated with Bisphenol A induced cognitive impairments in rat offspring. Birth Defects Res. 2021;113(12):911–24. https://doi.org/10.1002/bdr2.1886
28. Singha SP, Memon S, Bano U, Isaac AD, Shahani MY. Evaluation of p21 expression and related autism-like behavior in Bisphenol-A exposed offspring of Wistar albino rats. Birth Defects Res. 2022;114(11):536–50. https://doi.org/10.1002/bdr2.2022
29. Arbues A, Brees D, Chibout S-D, Fox T, Kammüller M, Portevin D. TNF-α antagonists differentially induce TGF-β1-dependent resuscitation of dormant-like Mycobacterium tuberculosis. PloS pathogens. 2020;16(2):e1008312. https://doi.org/10.1371/journal.ppat.1008312
30. Conrad A, Neven B, Mahlaoui N, Suarez F, Sokol H, Ruemmele FM, et al. Infections in patients with chronic granulomatous disease treated with tumor necrosis factor alpha blockers for inflammatory complications. J Clin Immunol. 2021;41(1):185-93. https://doi.org/10.1007/s10875-020-00901-8
31. Lopetuso LR, Cuomo C, Mignini I, Gasbarrini A, Papa A. Focus on anti-tumour necrosis factor (TNF)-α-related autoimmune diseases. Int J Mol Sci. 2023;24(9):8187. https://doi.org/10.3390/ijms24098187
32. Wadee R, Wadee AA. The pathology of lymphocytes, histiocytes, and immune mechanisms in Mycobacterium tuberculosis granulomas. Am J Trop Med Hyg. 2021;104(5):1796. https://doi.org/10.4269/ajtmh.20-1372
33. Shaikh P, Bano U, Memon S, Bano S, Gul S, Pirya. Neuroanatomical study of in-vivo brainstem abnormalities in autism spectrum disorder and their clinical correlations. Pak J Med Dent. 2025;14(3). https://doi.org/10.36283/ziun-pjmd14-3/039
34. Ghafoor A, Kashif S, Ali SMS, Memon S, Memon SS, Mehmood A, et al. Evaluating neurotherapeutic potential of naringenin by true experiments: insights into in-vivo psychiatry care models. Pak J Med Dent. 2025;14(3). https://doi.org/10.36283/ziun-pjmd14-3/043
35. Kim HW, Kim EH, Lee M, Jung I, Ahn SS. Risk of cancer, tuberculosis and serious infections in patients with ankylosing spondylitis, psoriatic arthritis and psoriasis treated with IL-17 and TNF-α inhibitors: a nationwide nested case-control analysis. Clin Exp Rheumatol. 2023;41(7):1491-9. https://doi.org/10.55563/clinexprheumatol/qkiorp
36. Mundra A, Yegiazaryan A, Karsian H, Alsaigh D, Bonavida V, Frame M, et al. Pathogenicity of type I interferons in mycobacterium tuberculosis. Int J Mol Sci. 2023;24(4):3919. https://doi.org/10.3390/ijms24043919
37. Petruccioli E, Farroni C, Cuzzi G, Vanini V, Palmieri F, Vittozzi P, et al. VIDAS® TB-IGRA reagents induce a CD4+ and CD8+ T-cell IFN-γ response for both TB infection and active TB. Int J Tuberc Lung Dis. 2022;26(1):65-8. https://doi.org/10.5588/ijtld.21.0478
38. Beig H, Singh A. Neutrophils from Protection to Pathogenesis in Tuberculosis. ACS Infect Dis. 2025;11(11):2926-50. https://doi.org/10.1021/acsinfecdis.5c00096
39. Park E-J, Kim I, Jo E-K. Host Immune Pathways to Mycobacterium Tuberculosis Infection. J Bacteriol Virol. 2024;54(3):167-90. https://doi.org/10.4167/jbv.2024.54.3.167
40. Madrid-Paulino E, Mata-Espinosa D, León-Contreras JC, Serrano-Fujarte I, Díaz de León-Guerrero S, Villaseñor T, et al. Klf10 favors Mycobacterium tuberculosis survival by impairing IFN-γ production and preventing macrophages reprograming to macropinocytosis. J Leukoc Biol. 2022;112(3):475-90. https://doi.org/10.1002/JLB.4MA0422-288R
41. Akter S, Chauhan KS, Dunlap MD, Choreño-Parra JA, Lu L, Esaulova E, et al. Mycobacterium tuberculosis infection drives a type I IFN signature in lung lymphocytes. Cell Rep. 2022;39(12):110983. https://doi.org/10.1016/j.celrep.2022.110983
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Shilpa Kishore, Abraham Mainaprem, Sher Rohan Paul (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





