Liquid Biopsy in Cancer Monitoring: Beyond Circulating Tumor DNA
Keywords:
Liquid Biopsy, Neoplasms, Circulating Tumor DNA, Extracellular Vesicles, RNA, Proteomics, High-Throughput Nucleotide SequencingAbstract
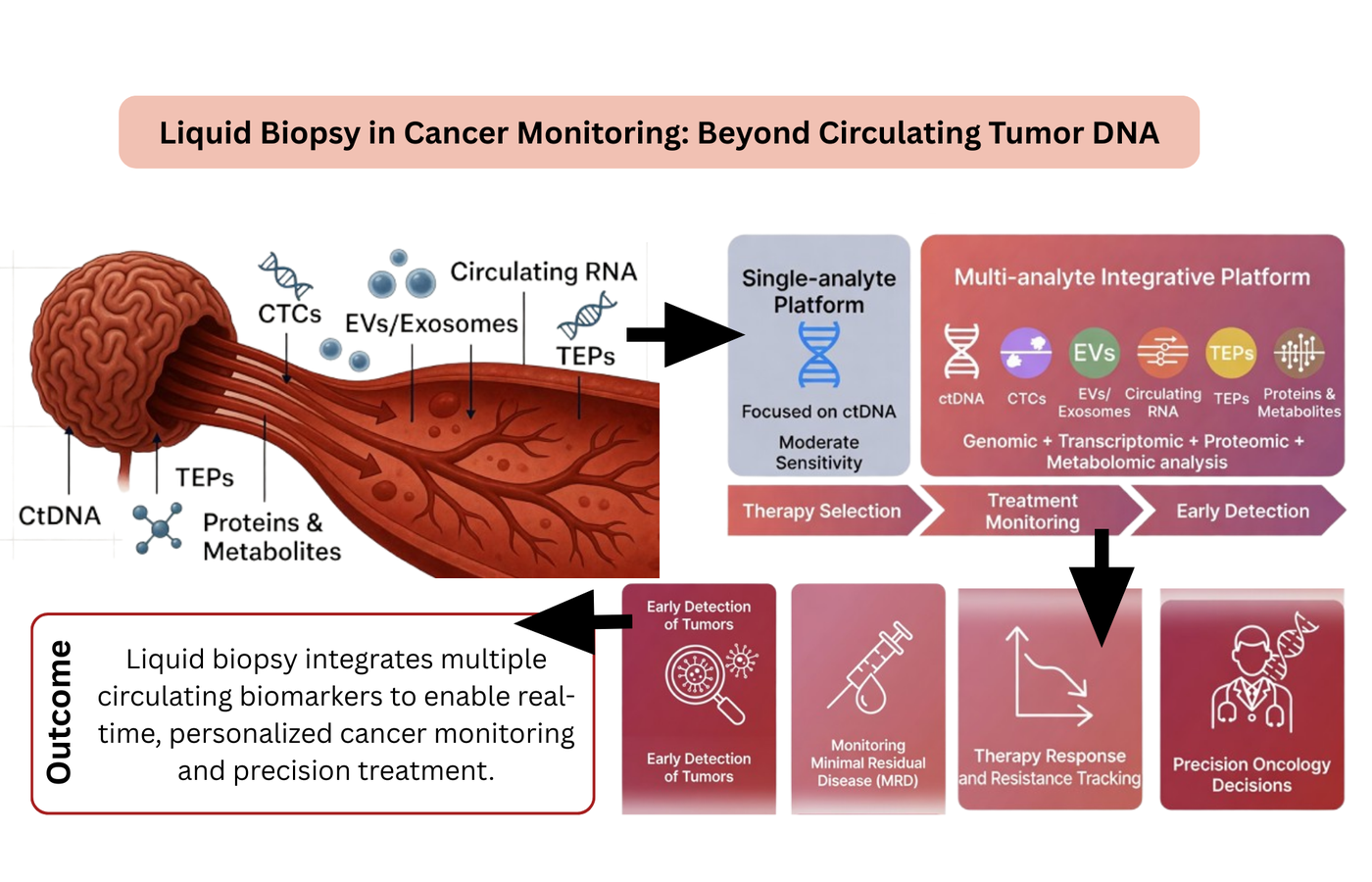
Liquid biopsy has become a potent, least invasive way of detecting, monitoring, and treating cancer by studying cancer-causing biomarkers in body fluids. The circulating tumor DNA (ctDNA) has long been the foundation of the liquid biopsy as it has the capability of measuring tumor-specific genetic changes. New developments have made it possible to identify and characterize a wide range of circulating analytes, such as ctDNA, extracellular vesicles, circulating RNA, tumor-educated platelets, and circulating proteins and metabolites. These biomarkers are associated with tumor viability, transcriptional activity, metastatic potential, and metabolic reprogramming. Innovations in next-generation sequencing, microfluidics, proteomics, and single-cell analysis have led to the detection platforms of many multi-analytes being immensely sensitive and reliable. Integrative liquid biopsy platforms can help to assess the evolution and therapeutic resistance of tumors in a holistic manner through the integration of genomic, transcriptomic, and proteomic and cellular data. Moreover, data interpretation has also been improved with the implementation of artificial intelligence and machine learning, resulting in reliable classification of the disease, risks stratification, and predict the response to treatment. Although significant progress has been made, complications such as, variability in analysis, absence of standardized protocols, high cost of operation, and biological heterogeneity, restrict the extensive clinical implementation. Technological advancement and interdisciplinary cooperation will continue to intensify the role of liquid biopsy in achieving the possibility of early detection, real-time monitoring, and optimal cancer management.
References
1. Santucci C, Carioli G, Bertuccio P, Malvezzi M, Pastorino U, Boffetta P, et al. Progress in cancer mortality, incidence, and survival: a global overview. Eur J Cancer Prev. 2020;29(5):367-81. https://doi.org/10.1097/CEJ.0000000000000594
2. Miller KD, Nogueira L, Devasia T, Mariotto AB, Yabroff KR, Jemal A, et al. Cancer treatment and survivorship statistics, 2022. Cancer J Clin. 2022;72(5):409-36. https://doi.org/10.3322/caac.21731
3. Howlader N, Forjaz G, Mooradian MJ, Meza R, Kong CY, Cronin KA, et al. The effect of advances in lung-cancer treatment on population mortality. N Engl J Med. 2020;383(7):640-9. https://doi.org/10.1056/NEJMoa1916623
4. Martins I, Ribeiro IP, Jorge J, Gonçalves AC, Sarmento-Ribeiro AB, Melo JB, et al. Liquid biopsies: applications for cancer diagnosis and monitoring. Genes. 2021;12(3):349. https://doi.org/10.3390/genes12030349
5. Li W, Liu J-B, Hou L-K, Yu F, Zhang J, Wu W, et al. Liquid biopsy in lung cancer: significance in diagnostics, prediction, and treatment monitoring. Mol Cancer. 2022;21(1):25. https://doi.org/10.1186/s12943-022-01505-z
6. Ma L, Guo H, Zhao Y, Liu Z, Wang C, Bu J, et al. Liquid biopsy in cancer: current status, challenges and future prospects. Signal Transduct Target Ther. 2024;9(1):336. https://doi.org/10.1038/s41392-024-02021-w
7. Lone SN, Nisar S, Masoodi T, Singh M, Rizwan A, Hashem S, et al. Liquid biopsy: a step closer to transform diagnosis, prognosis and future of cancer treatments. Mol Cancer. 2022;21(1):79. https://doi.org/10.1186/s12943-022-01543-7
8. Alix-Panabieres C. Perspective: The future of liquid biopsy. Nature. 2020;579(7800):S9-S. https://doi.org/10.1038/d41586-020-00844-5
9. Underwood JJ, Quadri RS, Kalva SP, Shah H, Sanjeevaiah AR, Beg MS, et al. Liquid biopsy for cancer: review and implications for the radiologist. Radiology. 2020;294(1):5-17. https://doi.org/10.1148/radiol.2019182584
10. Yu W, Hurley J, Roberts D, Chakrabortty S, Enderle D, Noerholm M, et al. Exosome-based liquid biopsies in cancer: opportunities and challenges. Ann Oncol. 2021;32(4):466-77. https://doi.org/10.1016/j.annonc.2021.01.074
11. Nikanjam M, Kato S, Kurzrock R. Liquid biopsy: current technology and clinical applications. J Hematol Oncol. 2022;15(1):131. https://doi.org/10.1186/s13045-022-01351-y
12. Goswami P, Memon S, Ujjan ID, Gul F. Altered placental expression of genes related to vitamin D metabolism and their correlation with spiral artery remodeling in preeclamptic women. Adv Life Sci. 2024;11(4):756–60. https://doi.org/10.62940/als.v11i4.1893
13. Szeto S, Kytölä S, Erkan EP, Ahtiainen M, Mecklin JP, Kuopio T, et al. Performance Comparison of Droplet Digital PCR and Next‐Generation Sequencing for Circulating Tumor DNA Detection in Non‐Metastatic Rectal Cancer. Cancer Med. 2025;14(9):e70943. https://doi.org/10.1002/cam4.70943
14. Galimberti S, Balducci S, Guerrini F, Del Re M, Cacciola R. Digital droplet PCR in hematologic malignancies: a new useful molecular tool. Diagnostics. 2022;12(6):1305. https://doi.org/10.3390/diagnostics12061305
15. Huerta M, Rosello S, Sabater L, Ferrer A, Tarazona N, Roda D, et al. Circulating tumor DNA detection by digital-droplet PCR in pancreatic ductal adenocarcinoma: a systematic review. Cancers. 2021;13(5):994. https://doi.org/10.3390/cancers13050994
16. Tan LL, Loganathan N, Agarwalla S, Yang C, Yuan W, Zeng J, et al. Current commercial dPCR platforms: technology and market review. Crit Rev Biotechnol. 2023;43(3):433-64. https://doi.org/10.1080/07388551.2022.2037503
17. Trouchet A, Gines G, Benhaim L, Taly V. Digital PCR: from early developments to its future application in clinics. Lab Chip. 2025;25(16):3921-61. https://doi.org/10.1039/d5lc00055f
18. Lin D, Shen L, Luo M, Zhang K, Li J, Yang Q, et al. Circulating tumor cells: biology and clinical significance. Signal Transduct Target Ther. 2021;6(1):404. https://doi.org/10.1038/s41392-021-00817-8
19. Mirabile A, Sangiorgio G, Bonacci PG, Bivona D, Nicitra E, Bonomo C, et al. Advancing pathogen identification: the role of digital PCR in enhancing diagnostic power in different settings. Diagnostics. 2024;14(15):1598. https://doi.org/10.3390/diagnostics14151598
20. Hurtado P, Martínez-Pena I, Piñeiro R. Dangerous liaisons: circulating tumor cells (CTCs) and cancer-associated fibroblasts (CAFs). Cancers. 2020;12(10):2861. https://doi.org/10.3390/cancers12102861
21. Lin Z, Luo G, Du W, Kong T, Liu C, Liu Z. Recent advances in microfluidic platforms applied in cancer metastasis: Circulating tumor cells'(CTCs) isolation and tumor‐on‐a‐chip. Small. 2020;16(9):1903899. https://doi.org/10.1002/smll.201903899
22. Di Bella MA. Overview and update on extracellular vesicles: considerations on exosomes and their application in modern medicine. Biology. 2022;11(6):804. https://doi.org/10.3390/biology11060804
23. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. https://doi.org/10.1126/science.aau6977
24. Mohammadipoor A, Hershfield MR, Linsenbardt HR, Smith J, Mack J, Natesan S, et al. Biological function of Extracellular Vesicles (EVs): a review of the field. Mol Biol Rep. 2023;50(10):8639-51. https://doi.org/10.1007/s11033-023-08624-w
25. Zhan C, Yang X, Yin X, Hou J. Exosomes and other extracellular vesicles in oral and salivary gland cancers. Oral Dis. 2020;26(5):865-75. https://doi.org/10.1111/odi.13172
26. Nasser JS, Altahoo N, Almosawi S, Alhermi A, Butler A. The role of MicroRNA, long non-coding RNA and circular RNA in the pathogenesis of polycystic ovary syndrome: a literature review. Int J Mol Sci. 2024;25(2):903. https://doi.org/10.3390/ijms25020903
27. Turai PI, Nyírő G, Butz H, Patócs A, Igaz P. MicroRNAs, long non-coding RNAs, and circular RNAs: potential biomarkers and therapeutic targets in pheochromocytoma/paraganglioma. Cancers. 2021;13(7):1522. https://doi.org/10.3390/cancers13071522
28. Nemeth K, Bayraktar R, Ferracin M, Calin G. Non-coding RNAs in disease: from mechanisms to therapeutics. Nat Rev Genet. 2024;25(3):211-32. https://doi.org/10.1038/s41576-023-00662-1
29. Roweth HG, Battinelli EM. Lessons to learn from tumor-educated platelets. Blood. 2021 Jun 10;137(23):3174-80. https://doi.org/10.1182/blood.2019003976
30. Varkey J, Nicolaides T. Tumor-educated platelets: a review of current and potential applications in solid tumors. Cureus. 2021;13(11).e19189. https://doi.org/10.7759/cureus.19189
31. Ding S, Dong X, Song X. Tumor educated platelet: the novel BioSource for cancer detection. Cancer Cell Int. 2023;23(1):91. https://doi.org/10.1186/s12935-023-02927-5
32. Donatti A, Canto AM, Godoi AB, da Rosa DC, Lopes-Cendes I. Circulating metabolites as potential biomarkers for neurological disorders—metabolites in neurological disorders. Metabolites. 2020;10(10):389. https://doi.org/10.3390/metabo10100389
33. Benson MD, Eisman AS, Tahir UA, Katz DH, Deng S, Ngo D, et al. Protein-metabolite association studies identify novel proteomic determinants of metabolite levels in human plasma. Cell Metab. 2023;35(9):1646-60. e3. https://doi.org/10.1016/j.cmet.2023.07.012
34. Lopez-Silva C, Surapaneni A, Schmidt IM, Upadhyay D, Srivastava A, Palsson R, et al. Circulating protein and metabolite correlates of histologically confirmed diabetic kidney disease. Kidney Med. 2024;6(12):100920. https://doi.org/10.1016/j.xkme.2024.100920
35. Hantikainen E, Weichenberger CX, Dordevic N, Verri Hernandes V, Foco L, Gögele M, et al. Metabolite and protein associations with general health in the population-based CHRIS study. 2024;14(1):26635. https://doi.org/10.1038/s41598-024-75627-3
36. Kilgour E, Rothwell DG, Brady G, Dive C. Liquid biopsy-based biomarkers of treatment response and resistance. Sci Rep. Cancer Cell. 2020;37(4):485-95. https://doi.org/10.1016/j.ccell.2020.03.012
37. Zieren RC, Zondervan PJ, Pienta KJ, Bex A, de Reijke TM, Bins AD. Diagnostic liquid biopsy biomarkers in renal cell cancer. Nat Rev Urol. 2024;21(3):133-57. https://doi.org/10.1038/s41585-023-00818-y
38. Zhang Z, Wu H, Chong W, Shang L, Jing C, Li L, et al. Liquid biopsy in gastric cancer: predictive and prognostic biomarkers. Cell Death Dis. 2022;13(10):903. https://doi.org/10.1038/s41419-022-05350-2
39. Park JH, Pyun WY, Park HW. Cancer metabolism: phenotype, signaling and therapeutic targets. Cells. 2020;9(10):2308. https://doi.org/10.3390/cells9102308
40. Raza A, Khan AQ, Inchakalody VP, Mestiri S, Yoosuf ZSK, Bedhiafi T, et al. Dynamic liquid biopsy components as predictive and prognostic biomarkers in colorectal cancer. J Exp Clin Cancer Res. 2022;41(1):99. https://doi.org/10.1186/s13046-022-02318-0
41. Toden S, Goel A. Non-coding RNAs as liquid biopsy biomarkers in cancer. Br J Cancer. 2022;126(3):351-60. https://doi.org/10.1038/s41416-021-01672-8
42. Lapitz A, Azkargorta M, Milkiewicz P, Olaizola P, Zhuravleva E, Grimsrud MM, et al. Liquid biopsy-based protein biomarkers for risk prediction, early diagnosis, and prognostication of cholangiocarcinoma. J Hepatol. 2023;79(1):93-108. https://doi.org/10.1016/j.jhep.2023.02.027
43. Koh DM, Papanikolaou N, Bick U, Illing R, Kahn Jr CE, Kalpathi-Cramer J, et al. Artificial intelligence and machine learning in cancer imaging. Commun Med. 2022;2(1):133. https://doi.org/10.1038/s43856-022-00199-0
44. Capobianco E. High-dimensional role of AI and machine learning in cancer research. Br J Cancer. 2022;126(4):523-32. https://doi.org/10.1038/s41416-021-01689-z
45. Hofmann L, Sallinger K, Haudum C, Smolle M, Heitzer E, Moser T, et al. A multi-analyte approach for improved sensitivity of liquid biopsies in prostate cancer. Cancers. 2020;12(8):2247. https://doi.org/10.3390/cancers12082247
46. Chen K, Sun J, Zhao H, Jiang R, Zheng J, Li Z, et al. Non-invasive lung cancer diagnosis and prognosis based on multi-analyte liquid biopsy. Mol Cancer. 2021;20(1):23. https://doi.org/10.1186/s12943-021-01323-9
47. Wade R, Nevitt S, Liu Y, Harden M, Khouja C, Raine G, et al. Multi-cancer early detection tests for general population screening: a systematic literature review. Health Technol Assess. 2025;29(2):1. https://doi.org/10.3310/DLMT1294
48. Held D, Bolland S, Freese R, Puskas R. A Protein-Based Blood Test for Multi-Cancer Diagnostics. Biomedicines. 2025;13(10):2510. https://doi.org/10.3390/biomedicines13102510
49. Heidrich I, Ačkar L, Mossahebi Mohammadi P, Pantel KJIJoC. Liquid biopsies: Potential and challenges. Int J Cancer. 2021;148(3):528-45. https://doi.org/10.1002/ijc.33217
50. Connal S, Cameron JM, Sala A, Brennan PM, Palmer DS, Palmer JD, et al. Liquid biopsies: the future of cancer early detection. J Transl Med. 2023;21(1):118. https://doi.org/10.1186/s12967-023-03960-8
51. Crocetto F, Russo G, Di Zazzo E, Pisapia P, Mirto BF, Palmieri A, et al. Liquid biopsy in prostate cancer management—Current challenges and future perspectives. Cancers. 2022;14(13):3272. https://doi.org/10.3390/cancers14133272
52. Chen YY, Han QY, Chen QY, Zhou WJ, Zhang JG, Zhang X, et al. Impact of sample processing and storage conditions on RNA quality of fresh-frozen cancer tissues. Biopreserv Biobank. 2023;21(5):510-7. https://doi.org/10.1089/bio.2022.0069
53. Su KY, Lee WL. Fourier transform infrared spectroscopy as a cancer screening and diagnostic tool: A review and prospects. Cancers. 2020;12(1):115. https://doi.org/10.3390/cancers12010115
54. Barkley D, Moncada R, Pour M, Liberman DA, Dryg I, Werba G, et al. Cancer cell states recur across tumor types and form specific interactions with the tumor microenvironment. Nat Genet. 2022;54(8):1192-201. https://doi.org/10.1038/s41588-022-01141-9
55. Kavan S, Kruse TA, Vogsen M, Hildebrandt MG, Thomassen MJC, Reviews M. Heterogeneity and tumor evolution reflected in liquid biopsy in metastatic breast cancer patients: a review. ancer Metastasis Rev. 2022;41(2):433-46. https://doi.org/10.1007/s10555-022-10023-9
56. Gristina V, Pepe F, Ogliari FR, Bazan Russo TD, Gottardo A, Russo G, et al. Smaller, cheaper, faster: where next for liquid biopsies?. Expert Rev Mol Diagn. 2025;25(9):555-73. https://doi.org/10.1080/14737159.2025.2534961
57. Alexandrou G, Mantikas KT, Allsopp R, Yapeter CA, Jahin M, Melnick T, et al. The evolution of affordable technologies in liquid biopsy diagnostics: the key to clinical implementation. Cancers. 2023;15(22):5434. https://doi.org/10.3390/cancers15225434
58. Memon S, Pratten MK. Developmental toxicity of ethanol in chick heart in ovo and in micromass culture can be prevented by addition of vitamin C and folic acid. Reprod Toxicol. 2009;28(2):262–9. https://doi.org/10.1016/j.reprotox.2009.03.018
59. Pratten M, Ahir BK, Smith-Hurst H, Memon S, Mutch P, Cumberland P. Primary cell and micromass culture in assessing developmental toxicity. Methods Mol Biol. 2012;889:115–46. https://doi.org/10.1007/978-1-61779-867-2_9
60. IJzerman MJ, de Boer J, Azad A, Degeling K, Geoghegan J, Hewitt C, et al. Towards routine implementation of liquid biopsies in cancer management: it is always too early, until suddenly it is too late. Diagnostics. 2021;11(1):103. https://doi.org/10.3390/diagnostics11010103
61. Hsu CC, Wu Y. Recent advances in nanotechnology-enabled biosensors for detection of exosomes as new cancer liquid biopsy. Exp Biol Med. 2022;247(23):2152-72. https://doi.org/10.1177/15353702221110813
62. Abusara OH, Agha ASA, Bardaweel SK. Advancements and innovations in liquid biopsy through microfluidic technology for cancer diagnosis. Analyst. 2025;150(9):1711-25. https://doi.org/10.1039/d5an00105f
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Hajra Ahmad, Irshad Ali, Mubashra Khadim (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





