Molecular and Cytopathological Determinants of Malignant Transformations in Oral Leukoplakia: A Meta-Analytic Synthesis of AgNOR, DNA Ploidy, Cytokeratin, and Podoplanin Evidence
Keywords:
Oral Leukoplakia, Oral Neoplasms, Epithelial Dysplasia, Biomarkers, Tumor, Disease ProgressionAbstract
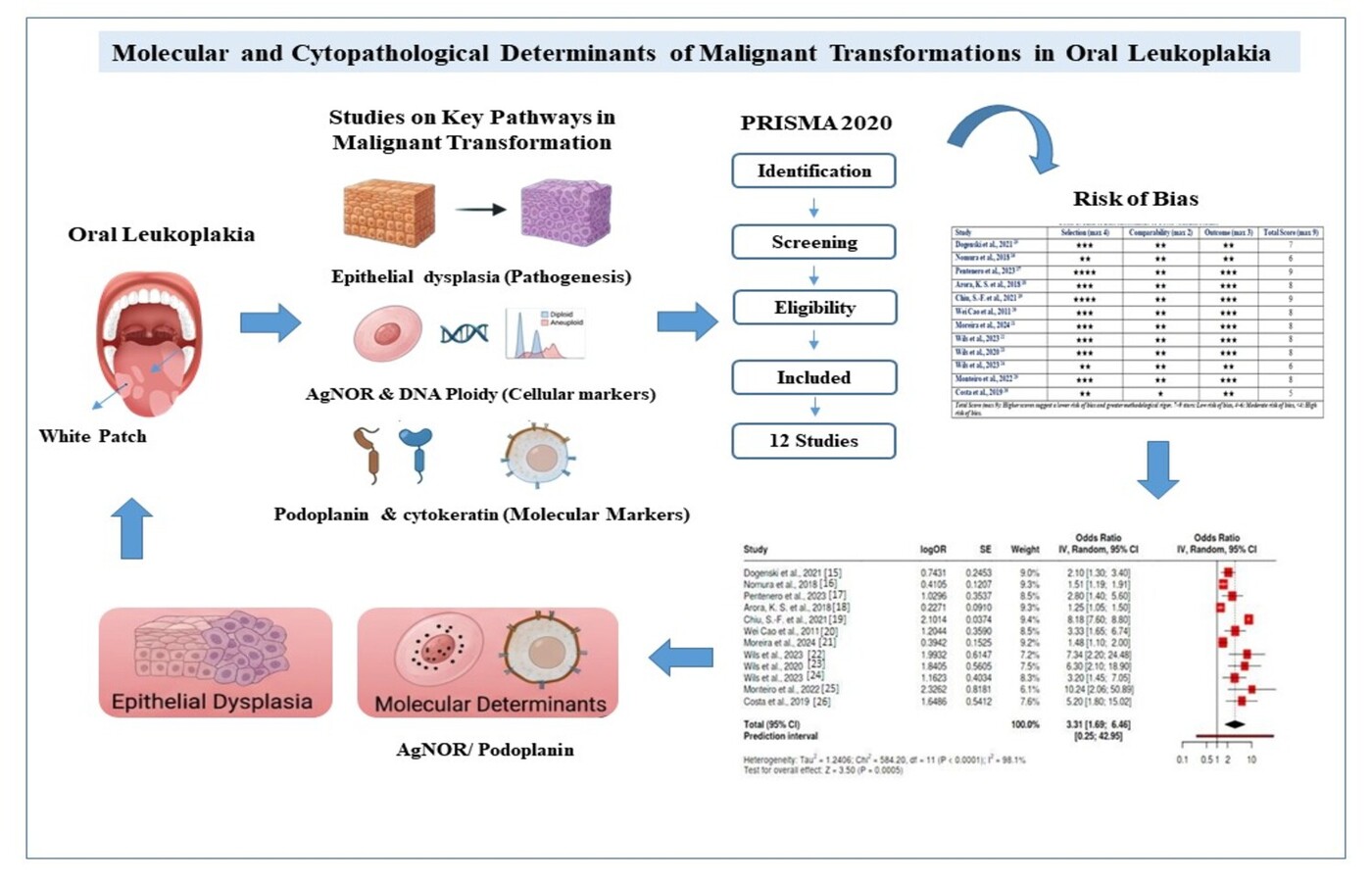
Background: The diagnosis of Oral leukoplakia exists as one of several potentially cancer-causing oral disorders that frequently precede the development of oral cancer. Such lesions occur as white tissue with an unknown origin and demonstrate various degrees of dysplastic change in the epithelial cells. The dysplastic condition functions as a critical component that leads to cancer formation. The research reviews diagnostic features and prognostic indicators of leukoplakia, which contributes to cancer transformation by examining dysplasia categories and lesional characteristics. Methodology: The research adopted a thorough database search in PubMed, Scopus, and Web of Science according to PRISMA protocols. Observational studies, together with retrospective cohort research, evaluated oral malignancy from leukoplakia. Researchers studied three main outcome groups, including clinical indicators as well as histopathological features, together with molecular data. The Newcastle-Ottawa Scale was used for evaluating study quality. Results: Research findings showed that epithelial dysplasia, together with AgNOR count and DNA ploidy analysis, and biomarker expression tests, all demonstrated meaningful connections to malignant cell transformation risk markers. Research has determined that CK13/17 expression and podoplanin measurement levels act as indicators that help predict disease progression. The study results demonstrated that specific markers delivered steady evidence about their connection with increased cancer cell transformation frequency. The reliability ratings of most studies were measured as moderate to high through their risk of bias assessment. Conclusion: A combined analysis of tissue structure examination and molecular data improves the ability to diagnose leukoplakia as an oral malignancy indicator and enhances risk estimation.
References
1. Kumari P, Debta P, Dixit A. Oral potentially malignant disorders: Etiology, pathogenesis, and transformation into oral cancer. Front Pharmacol. 2022 Mar;13:825266. https://doi.org/10.3389/fphar.2022.825266
2. Brouns E, Baart J, Karagozoglu K, Aartman I, Bloemena E, van der Waal I. Malignant transformation of oral leukoplakia in a well-defined cohort of 144 patients. Oral Dis. 2014 Mar;20(3):e19-24. https://doi.org/10.1111/odi.12095
3. Das NK, Kadir AKMS, Shemanto MU, Akhter E, Sharfaraz A, Tripura S, et al. Genetic revelation of the potentially malignant disorders in the oral and maxillofacial region. In: Population Genetics—From DNA to Evolutionary Biology. IntechOpen; 2023. https://doi.org/10.5772/intechopen.112697
4. Elimairi I, Sami A, Yousef B. Oral cancer and potentially malignant disorders. In: Histopathology—An Update. IntechOpen; 2017. https://doi.org/10.5772/intechopen.71887
5. Gómez-Armayones S, Chimenos-Küstner E, Arranz C, Tous S, Marquez S, Penín RM, et al. Risk factors for oral epithelial dysplasias to become malignant: Clinical implications. Int J Oral Maxillofac Surg. 2022 Apr;51(4):473-480. https://doi.org/10.1016/j.ijom.2021.08.012
6. Gupta I, Badrzadeh F, Tsentalovich Y, Gaykalova DA. Connecting the dots: Investigating the link between environmental, genetic, and epigenetic influences in metabolomic alterations in oral squamous cell carcinoma. J Exp Clin Cancer Res. 2024 Jan;43(1):239. https://doi.org/10.1186/s13046-024-03141-5
7. Alam SS, Ahmed S, Farook TH, Dudley J. Predicting leukoplakia and oral squamous cell carcinoma using interpretable machine learning: A retrospective analysis. Oral. 2024;4(3):32. https://doi.org/10.3390/oral4030032
8. He S, Chakraborty R, Ranganathan S. Proliferation and apoptosis pathways and factors in oral squamous cell carcinoma. Int J Mol Sci. 2022 Mar;23(3):1562. https://doi.org/10.3390/ijms23031562
9. Sundaram E, Pal US, Sowmya MV, Kumar V, Yadav S. Field cancerisation in oral squamous cell carcinoma patients: A systematic review. J Maxillofac Oral Surg. 2025 Apr;24(2):416–431. https://doi.org/10.1007/s12663-024-02287-1
10. Jing F, Zhang J, Cai X, Zhou X, Bai J, Zhang H, et al. Screening for biomarkers for progression from oral leukoplakia to oral squamous cell carcinoma and evaluation of diagnostic efficacy by multiple machine learning algorithms. Cancers. 2022 Dec;14(23):5808. https://doi.org/10.3390/cancers14235808
11. Khong B, Ferlito S, Quek S, Conte G, Ingrassia A, Lechien JR, et al. Past, present, and future diagnostic methods for the early noninvasive detection of oral premalignant lesions: A state of the art and systematic review. Ear Nose Throat J. 2024 Apr;01455613241245204. https://doi.org/10.1177/01455613241245204
12. Parums DV. Review articles, systematic reviews, meta-analysis, and the updated preferred reporting items for systematic reviews and meta-analyses (PRISMA) 2020 guidelines. Med Sci Monit. 2021 Aug;27:e934475-1. https://doi.org/10.12659/MSM.934475
13. Carra MC, Romandini P, Romandini M. Risk of bias evaluation of cross-sectional studies: Adaptation of the Newcastle–Ottawa Scale. J Periodontal Res. 2025 Apr;60(2):e13405. https://doi.org/10.1111/jre.13405
14. Jun S, Park H, Kim UJ, Lee HA, Park B, Lee SY, et al. The combined effects of alcohol consumption and smoking on cancer risk by exposure level: A systematic review and meta-analysis. J Korean Med Sci. 2024 Jun;39(22):e185. https://doi.org/10.3346/jkms.2024.39.e185
15. Dogenski LC, de Figueiredo Ribeiro S, Gambin DJ, Maso PC, Linden MSS, Trentin MS, et al. Oral leukoplakia-epidemiological survey and histochemical analysis of 107 cases in Brazil. Clin Oral Investig. 2021 Apr;25(4):1859-1867. https://doi.org/10.1007/s00784-020-03488-x
16. Nomura H, Sakamoto K, Sugihara T, Okamoto S, Aoki Y, Tanigawa T, et al. Oral leukoplakia, a precancerous lesion of squamous cell carcinoma, in patients with long-term pegylated liposomal doxorubicin treatment. Medicine. 2018 Feb;97(7):e9932. https://doi.org/10.1097/MD.0000000000009932
17. Pentenero M, Castagnola P, Castillo FV, Isaevska E, Sutera S, Gandolfo S. Predictors of malignant transformation in oral leukoplakia and proliferative verrucous leukoplakia: An observational prospective study including the DNA ploidy status. Head Neck. 2023 Oct;45(10):2589-2604. https://doi.org/10.1002/hed.27483
18. Arora KS, Bansal R, Mohapatra S, Verma A, Sharma S, Pareek S. Prevention of malignant transformation of oral leukoplakia and oral lichen planus using laser: An observational study. Asian Pac J Cancer Prev. 2018 Dec;19(12):3635-3641. https://doi.org/10.31557/APJCP.2018.19.12.3635
19. Chiu SF, Ho CH, Chen YC, Wu LW, Chen YL, Wu JH, et al. Malignant transformation of oral potentially malignant disorders in Taiwan: An observational nationwide population database study. Medicine. 2021 Sep;100(9):e24934. https://doi.org/10.1097/MD.0000000000024934
20. Cao W, Younis RH, Li J, Chen H, Xia R, Mao L, et al. EZH2 promotes malignant phenotypes and is a predictor of oral cancer development in patients with oral leukoplakia. Cancer Prev Res. 2011 Nov;4(11):1816-1824. https://doi.org/10.1158/1940-6207.CAPR-11-0130
21. Moreira MD, Maia FD, Zimbrão VL, Collodetti E, Grão-Velloso TR, Pimenta-Barros LA, et al. Demographic and clinicopathological comparison among oral lichen planus, lichenoid lesions and proliferative verrucous leukoplakia: A retrospective study. BMC Oral Health. 2024 Jan;24(1):1512. https://doi.org/10.1186/s12903-024-05305-3
22. Wils LJ, Poell JB, Brink A, Evren I, Brouns ER, de Visscher JGAM, et al. Elucidating the genetic landscape of oral leukoplakia to predict malignant transformation. Clin Cancer Res. 2023 Feb;29(3):602-613. https://doi.org/10.1158/1078-0432.CCR-22-2210
23. Wils LJ, Poell JB, Evren I, Koopman MS, Brouns EREA, de Visscher JGAM, et al. Incorporation of differentiated dysplasia improves prediction of oral leukoplakia at increased risk of malignant progression. Mod Pathol. 2020 Jun;33(6):1033-1040. https://doi.org/10.1038/s41379-019-0444-0
24. Wils LJ, Poell JB, Peferoen LAN, Evren I, Brouns ER, de Visscher JGAM, et al. The role of differentiated dysplasia in the prediction of malignant transformation of oral leukoplakia. J Oral Pathol Med. 2023 Oct;52(10):930-938. https://doi.org/10.1111/jop.13483
25. Monteiro L, do Amaral B, Delgado L, Garcês F, Salazar F, Pacheco JJ, et al. Podoplanin expression independently and jointly with oral epithelial dysplasia grade acts as a potential biomarker of malignant transformation in oral leukoplakia. Biomolecules. 2022 May;12(5):606. https://doi.org/10.3390/biom12050606
26. Herrera Costa F, Narana Ribeiro El Achkar V, Costa V, Paladini I, Kowalski LP, Rodarte Carvalho Y, et al. Different expression of aldehyde dehydrogenases 1A1 and 2 in oral leukoplakia with epithelial dysplasia and in oral squamous cell carcinoma. Appl Immunohistochem Mol Morphol. 2019 Jul;27(7):537-542. https://doi.org/10.1097/PAI.0000000000000612
27. Lin HJ, Wang XL, Tian MY, Li XL, Tan HZ. Betel quid chewing and oral potential malignant disorders and the impact of smoking and drinking: A meta-analysis. World J Clin Cases. 2022 Mar;10(10):3131-3142. https://doi.org/10.12998/wjcc.v10.i10.3131
28. Nasser W, Flechtenmacher C, Holzinger D, Hofele C, Bosch FX. Aberrant expression of p53, p16INK4a and Ki-67 as basic biomarker for malignant progression of oral leukoplakias. J Oral Pathol Med. 2011 Aug;40(8):629-635. https://doi.org/10.1111/j.1600-0714.2011.01026.x
29. Ranganathan K, Kavitha L. Oral epithelial dysplasia: Classifications and clinical relevance in risk assessment of oral potentially malignant disorders. J Oral Maxillofac Pathol. 2019 Jan;23(1):19-27. https://doi.org/10.4103/jomfp.JOMFP_13_19
30. Reibel J. Prognosis of oral pre-malignant lesions: Significance of clinical, histopathological, and molecular biological characteristics. Crit Rev Oral Biol Med. 2003 Jan;14(1):47-62. https://doi.org/10.1177/154411130301400105
31. Rivero LF, Graudenz MS, Aschton-Prolla P, Delgado AM, Kliemann LM. Accuracy of p53 and ki-67 in the graduation of phyllodes tumor, a model for practical application. Surg Exp Pathol. 2020 Mar;3(1):7. https://doi.org/10.1186/s42047-020-0058-3
32. Sankaranarayanan R, Ramadas K, Amarasinghe H, Subramanian S, Johnson N. Oral cancer: Prevention, early detection, and treatment. In: Gelband H, Jha P, Sankaranarayanan R, Horton S, editors. Cancer: Disease Control Priorities. 3rd ed. The World Bank; 2015. https://doi.org/10.1596/978-1-4648-0349-9
33. Sanz-García A, Pérez-Romero M, Pastor J, Sola RG, Vega-Zelaya L, Vega G, et al. Potential EEG biomarkers of sedation doses in intensive care patients unveiled by using a machine learning approach. J Neural Eng. 2019 Feb;16(2):026031. https://doi.org/10.1088/1741-2552/ab039f
34. Bernard C, Zhang JZ, Klieb H, Blanas N, Xu W, Magalhaes M. Clinical outcomes of oral epithelial dysplasia managed by observation versus excision. Head Neck. 2023 Dec;45(12):3096–3106. https://doi.org/10.1002/hed.27539
35. Tan Y, Wang Z, Xu M, Li B, Huang Z, Qin S, et al. Oral squamous cell carcinomas: State of the field and emerging directions. Int J Oral Sci. 2023 Mar;15(1):1-23. https://doi.org/10.1038/s41368-023-00249-w
36. Van Zyl AW, van Heerden MB, Langenegger E, van Heerden WFP. Correlation between dysplasia and ploidy status in oral leukoplakia. Head Neck Pathol. 2012 Sep;6(3):322-327. https://doi.org/10.1007/s12105-012-0352-9
37. Villa A, Woo SB. Leukoplakia—A diagnostic and management algorithm. J Oral Maxillofac Surg. 2017 Apr;75(4):723-734. https://doi.org/10.1016/j.joms.2016.10.012
38. You JR, Chen YT, Hsieh CY, Chen SY, Lin TY, Shih JS, et al. Exploring possible diagnostic precancerous biomarkers for oral submucous fibrosis: A narrative review. Cancers. 2023 Oct;15(19):4812. https://doi.org/10.3390/cancers15194812
39. Zygogianni AG, Kyrgias G, Karakitsos P, Psyrri A, Kouvaris J, Kelekis N, et al. Oral squamous cell cancer: Early detection and the role of alcohol and smoking. Head Neck Oncol. 2011 Jan;3:2. https://doi.org/10.1186/1758-3284-3-2
40. Chaturvedi AK, Udaltsova N, Engels EA, Katzel JA, Yanik EL, Katki HA, Lingen MW, Silverberg MJ. Oral leukoplakia and risk of progression to oral cancer: a population-based cohort study. J Natl Cancer Inst. 2020 Oct;112(10):1047–1054. https://doi.org/10.1093/jnci/djz238
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Kashaf Asghar, Rameesha Ashraf, Muhammad Taimoor, Aqeel Ahmed, Hamdan Khalid (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





