Exploring the Role of Oxidative Stress-Related Molecular Biomarkers in Cardiovascular Disease Progression and Therapeutic Modulations: A Systematic Analysis
Keywords:
Oxidative Stress, Reactive Oxygen Intermediates, Superoxide Dismutase, Malondialdehyde, Endothelial, Hypertension, Cardiovascular Disease, Antioxidant EffectsAbstract
ABSTRACT
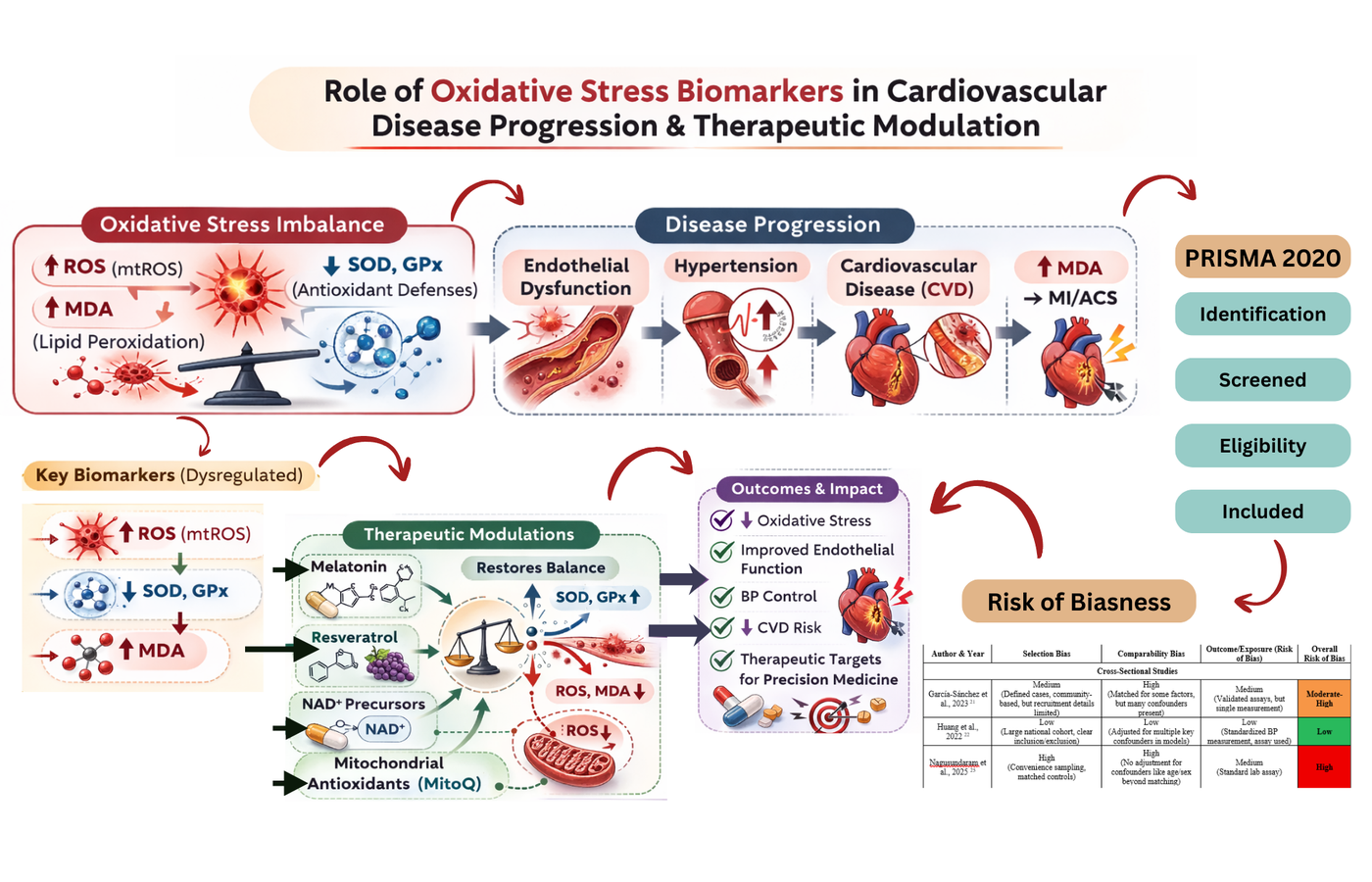
Background: Oxidative stress plays a significant role in the pathogenesis of cardiovascular diseases (CVD), endothelial dysfunction and hypertension. The purpose of this study was to synthesize the evidence on the role of critical oxidative stress biomarkers in the etiology of associated diseases to determine their therapeutic potential. Methods: PRISMA 2020 guidelines were followed to conduct this study. Reported literature was searched using PubMed, Scopus, Web of Science, and Google Scholar from 2014 to 2025. Those studies that investigated certain biomarkers of oxidative stress (ROS,), glutathione peroxidase (GPx), malondialdehyde (MDA)) in the pathophysiology of CVD, endothelial dysfunction and hypertension were included. Non-English articles and studies without mechanistic or therapeutic outcomes were excluded. The risk of bias was evaluated with the help of Newcastle-Ottawa Scale, Cochrane risk of bias-2, and SYRCLE tool, and certainty of evidence was measured with help of the GRADE approach. Results: Findings of twelve studies were included in this review, which demonstrated that high level of mitochondrial ROS and MDA, and low activity of SOD and GPx were major contributor to endothelial dysfunction, hypertension and CVD. Melatonin, resveratrol, NAD precursors and mitochondrial-targeted antioxidants proved to be effective therapeutic agents. Risks of bias among included studies ranged from low to high, and the confidence of the evidence ranged from low to moderate. Conclusion: Oxidative stress biomarkers are critical in cardiovascular disease pathogenesis and hold significant therapeutic potential. Further extensive trials are needed to validate these indicators as diagnostic tools and convert targeted antioxidant therapy into successful precision medicine.
References
1. Donia T, Khamis A. Management of oxidative stress and inflammation in cardiovascular diseases: mechanisms and challenges. Environ Sci Pollut Res. 2021;28(26):34121-53. https://doi.org/10.1007/s11356-021-14109-9
2. Scioli MG, Storti G, D’Amico F, Rodríguez Guzmán R, Centofanti F, Doldo E, et al. Oxidative stress and new pathogenetic mechanisms in endothelial dysfunction: potential diagnostic biomarkers and therapeutic targets. J Clin Med. 2020;9(6):1995. https://doi.org/10.3390/jcm9061995
3. Higashi Y. Roles of oxidative stress and inflammation in vascular endothelial dysfunction-related disease. Antioxidants. 2022;11(10):1958. https://doi.org/10.3390/antiox11101958
4. Sies H, Belousov VV, Chandel NS, Davies MJ, Jones DP, Mann GE, et al. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat Rev Mol Cell Biol. 2022;23(7):499-515. https://doi.org/10.1038/s41580-022-00456-z
5. Gianazza E, Brioschi M, Martinez Fernandez A, Casalnuovo F, Altomare A, Aldini G, et al. Lipid peroxidation in atherosclerotic cardiovascular diseases. Antioxid Redox Signal. 2021;34(1):49-98. https://doi.org/10.1089/ars.2019.7955
6. García-Sánchez A, Miranda-Díaz AG, Cardona-Muñoz EG. The role of oxidative stress in physiopathology and pharmacological treatment with pro‐and antioxidant properties in chronic diseases. Oxid Med Cell Longev. 2020;2020(1):2082145. https://doi.org/10.1155/2020/2082145
7. Reddy VP. Oxidative stress in health and disease. Biomedicines. 2023;11(11):2925. https://doi.org/10.3390/biomedicines11112925
8. Panda P, Verma HK, Lakkakula S, Merchant N, Kadir F, Rahman S, et al. Biomarkers of oxidative stress tethered to cardiovascular diseases. Oxid Med Cell Longev. 2022;2022(1):9154295. https://doi.org/10.1155/2022/9154295
9. Ait Tayeb AEK, Poinsignon V, Chappell K, Bouligand J, Becquemont L, Verstuyft C. Major depressive disorder and oxidative stress: a review of peripheral and genetic biomarkers according to clinical characteristics and disease stages. Antioxidants. 2023;12(4):942. https://doi.org/10.3390/antiox12040942
10. Sami AJ, Khalid M, Nasar S, Mangat HA, Butt YN. Sustained release of ibuprofen by a novel formulated hydrogel containing graphene oxide. J Mol Pharm Org Process Res. 2018;6:143. https://doi.org/10.4172/2329-9053.1000143
11. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. bmj. 2021;372. https://doi.org/10.1136/bmj.n71
12. Hooijmans CR, Rovers MM, De Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14(1):43. https://doi.org/10.1186/1471-2288-14-43
13. Carra MC, Romandini P, Romandini M. Risk of Bias Evaluation of Cross‐Sectional Studies: Adaptation of the Newcastle‐Ottawa Scale. J Periodontal Res. 2025. https://doi.org/10.1111/jre.13405
14. Moore TH, Higgins JP, Dwan K. Ten tips for successful assessment of risk of bias in randomized trials using the RoB 2 tool: Early lessons from Cochrane. Cochrane Evid Synth Methods. 2023;1(10):e12031. https://doi.org/10.1002/cesm.12031
15. Li P, Xie C, Zhong J, Guo Z, Guo K, Tu Q. Melatonin attenuates ox‐LDL‐Induced endothelial dysfunction by reducing ER stress and inhibiting JNK/Mff signaling. Oxid Med Cell Longev. 2021;2021(1):5589612. https://doi.org/10.1155/2021/5589612
16. Cao X, Wu Y, Hong H, Tian XY. Sirtuin 3 dependent and independent effects of NAD+ to suppress vascular inflammation and improve endothelial function in mice. Antioxidants. 2022;11(4):706. https://doi.org/10.3390/antiox11040706
17. Zhou X, Chen M, Zeng X, Yang J, Deng H, Yi L, et al. Resveratrol regulates mitochondrial reactive oxygen species homeostasis through Sirt3 signaling pathway in human vascular endothelial cells. Cell Death Dis. 2014;5(12):e1576-e. https://doi.org/10.1038/cddis.2014.530
18. Murray KO, Ludwig KR, Darvish S, Coppock ME, Seals DR, Rossman MJ. Chronic mitochondria antioxidant treatment in older adults alters the circulating milieu to improve endothelial cell function and mitochondrial oxidative stress. Am J Physiol Heart Circ Physiol. 2023;325(1):H187-H94. https://doi.org/10.1152/ajpheart.00270.2023
19. He J, Liu X, Su C, Wu F, Sun J, Zhang J, et al. Inhibition of mitochondrial oxidative damage improves reendothelialization capacity of endothelial progenitor cells via SIRT3 (Sirtuin 3)-enhanced SOD2 (superoxide dismutase 2) deacetylation in hypertension. Arterioscler Thromb Vasc Biol. 2019;39(8):1682-98. https://doi.org/10.1161/ATVBAHA.119.312613
20. Dikalova AE, Pandey A, Xiao L, Arslanbaeva L, Sidorova T, Lopez MG, et al. Mitochondrial deacetylase Sirt3 reduces vascular dysfunction and hypertension while Sirt3 depletion in essential hypertension is linked to vascular inflammation and oxidative stress. Circ Res. 2020;126(4):439-52. https://doi.org/10.1161/CIRCRESAHA.119.315767
21. García-Sánchez A, Gómez-Hermosillo L, Casillas-Moreno J, Pacheco-Moisés F, Campos-Bayardo TI, Román-Rojas D, et al. Prevalence of hypertension and obesity: profile of mitochondrial function and markers of inflammation and oxidative stress. Antioxidants. 2023;12(1):165. https://doi.org/10.3390/antiox12010165
22. Huang Y, Wei L, Hong C, Qi-Fan L, Ju-Xiang L, Jin-Zhu H. The L-shaped association between superoxide dismutase levels and blood pressure in older Chinese adults: community-based, cross-sectional study. J Geriatr Cardiol. 2022;19(1):71. https://doi.org/10.11909/j.issn.1671-5411.2022.01.002
23. Moronkeji A, Olaniyan M. Evaluation of 8-hydroxy-2'-deoxyguanosine, interleukin-8 and malondialdehyde among patients with cardiovascular diseases in Ibadan, Oyo State. Sokoto J Med Lab Sci. 2024;9(2):160-9. https://doi.org/10.11909/j.issn.1671-5411.2022.01.002
24. Amioka N, Miyoshi T, Otsuka H, Yamada D, Takaishi A, Ueeda M, et al. Serum malondialdehyde-modified low-density lipoprotein levels on admission predict prognosis in patients with acute coronary syndrome undergoing percutaneous coronary intervention. J Cardiol. 2019;74(3):258-66. https://doi.org/10.1016/j.jjcc.2019.02.012
25. Nagasundaram M, Singh YR, Singh TSD, Shaini L, Laishram N. Study Of Serum Malondialdehyde Level In Patients with Acute Myocardial Infarction (AMI). Int J Acad Med Pharm. 2025;7(3):625-9. https://doi.org/10.47009/jamp.2025.7.3.120
26. Arif MA, Ahmeid MS, Allaw SA. Malondialdehyde level in the patients subjected to open heart surgery in association with lipid profile. Mustansiriya Med J. 2019;18(1):30-5. https://doi.org/10.4103/MJ.MJ_34_18
27. Minjares M, Wu W, Wang J-M. Oxidative stress and microRNAs in endothelial cells under metabolic disorders. Cells. 2023;12(9):1341. https://doi.org/10.3390/cells12091341
28. Radovanović J, Banjac K, Obradović MM, Isenović ER. Antioxidant enzymes and vascular diseases. Explor Med. 2021;2(6):544-55. https://doi.org/10.37349/emed.2021.00070
29. Qazi N, Memon S, Memon F, Goswami P, Sirhandi BR, Goswami B. Antioxidant and hepato-protective effects of ginger in comparison with atorvastatin in hyperlipidemic albino mice. JMMC. 2024 Oct 6;15(1):21-26. https://doi.org/10.62118/jmmc.v15i1.464
30. Bano U, Memon S, Shahani MY, Shaikh P, Gul S. Epigenetic effects of in utero bisphenol A administration: Diabetogenic and atherogenic changes in mice offspring. Iran J Basic Med Sci. 2019 May;22(5):521-528. https://doi.org/10.22038/ijbms.2019.29909.7357
31. de Paula da Silva MV, Villar-Delfino PH, Nogueira-Machado JA, Volpe CM. IL-6, IL-1β, and MDA Correlate with Thrombolysis in Myocardial Infarction (TIMI) Risk Score in Patients with Acute Coronary Syndrome. Recent Adv Inflamm Allergy Drug Discov. 2021;15(2):71-9. https://doi.org/10.2174/2772270816666220211091231
32. Brohi GH, Memon SR, Shahani MY, Memon S, Bano U. Acute myocardial infarction: short-term effect of successful aspiration in primary stenting on patients with acute myocardial infarction. Prof Med J. 2018;25(8):1223–8. https://doi.org/10.29309/TPMJ/2018.25.08.44
33. Shahani SB, Shaikh P, Memon SG, Memon S. Possible prevention of reactive oxygen species induced human trabecular meshwork cell damage by resveratrol and ascorbic acid. Prof Med J. 2019 May 31;26(07):1036-1041. https://doi.org/10.29309/TPMJ/2019.26.07.3229
34. Liu Y, Huang Y, Xu C, An P, Luo Y, Jiao L, et al. Mitochondrial dysfunction and therapeutic perspectives in cardiovascular diseases. Int J Mol Sci. 2022;23(24):16053. https://doi.org/10.3390/ijms232416053
35. Li H, Hu Q, Zhu D, Wu D. The Role of NAD+ Metabolism in Cardiovascular Diseases: Mechanisms and Prospects. Am J Cardiovasc Drugs. 2025;25(3):307-27. https://doi.org/10.1007/s40256-024-00711-y
36. Mladenov M, Lubomirov L, Grisk O, Avtanski D, Mitrokhin V, Sazdova I, et al. Oxidative stress, reductive stress and antioxidants in vascular pathogenesis and aging. Antioxidants. 2023;12(5):1126. https://doi.org/10.3390/antiox12051126
37. Memon SG, Shaikh P, Shahani MY, Bano U, Rani S, Memon S. Possible protection against cisplatin induced behavioral changes by dietary antioxidants in adult albino mice. Prof Med J. 2020;27(6):1217–23. https://doi.org/10.29309/TPMJ/2020.27.06.4162
38. Salnikova D, Orekhova V, Grechko A, Starodubova A, Bezsonov E, Popkova T, et al. Mitochondrial dysfunction in vascular wall cells and its role in atherosclerosis. International Int J Mol Sci. 2021;22(16):8990. https://doi.org/10.3390/ijms22168990
39. Beg MA, Huang M, Vick L, Rao KS, Zhang J, Chen Y. Targeting mitochondrial dynamics and redox regulation in cardiovascular diseases. Trends Pharmacol Sci. 2024;45(4):290-303. https://doi.org/10.1016/j.tips.2024.02.001
40. Aladağ N, Asoğlu R, Ozdemir M, Asoğlu E, Derin AR, Demir C, et al. Oxidants and antioxidants in myocardial infarction (MI): Investigation of ischemia modified albumin, malondialdehyde, superoxide dismutase and catalase in individuals diagnosed with ST elevated myocardial infarction (STEMI) and non-STEMI (NSTEMI). J Med Biochem. 2021;40(3):286. https://doi.org/10.5937/jomb0-28879
41. Yan Q, Liu S, Sun Y, Chen C, Yang S, Lin M, et al. Targeting oxidative stress as a preventive and therapeutic approach for cardiovascular disease. J Transl Med. 2023;21(1):519. https://doi.org/10.1186/s12967-023-04361-7
42. Güler MC, Tanyeli A, Akdemir FNE, Eraslan E, Şebin SÖ, Erdoğan DG, et al. An overview of ischemia–reperfusion injury: review on oxidative stress and inflammatory response. Eurasian J Med. 2022;54(Suppl 1):S62. https://doi.org/10.5152/eurasianjmed.2022.22293
43. Soni SK, Basu P, Singaravel M, Sharma R, Pandi-Perumal SR, Cardinali DP, et al. Sirtuins and the circadian clock interplay in cardioprotection: focus on sirtuin 1. Cell Mol Life Sci. 2021;78(6):2503-15. https://doi.org/10.1007/s00018-020-03713-6
44. Karakayali M, Öğün M, Artaç İ, Omar T, İliş D, Arslan A, et al. Inducible nitric oxide synthase (iNOS) is a potential marker of myocardial infarction with Non-obstructive coronary artery disease (MINOCA). Bağcılar Tıp Bülteni. 2024;9(3):188-95. https://doi.org/10.4274/BMB.galenos.2024.2023-12-107
45. Zhao H, Zhang R, Yan X, Fan K. Superoxide dismutase nanozymes: an emerging star for anti-oxidation. J Mater Chem. 2021;9(35):6939-57. https://doi.org/10.1039/d1tb00720c
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Siddiqa Fatima, Jannat Ikram, Sadia Akram, Palwisha Dar (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





