Perioperative Modulation of Recovery Kinetics and Analgesic Efficiency in Minimally Invasive Abdominal Surgery
Keywords:
Post-operative Period, Analgesics, Opioid, Recovery of Function, Laparoscopy, Anesthesia, General Pain Measurement, Enhanced Recovery After Surgery (ERAS) ProtocolsAbstract
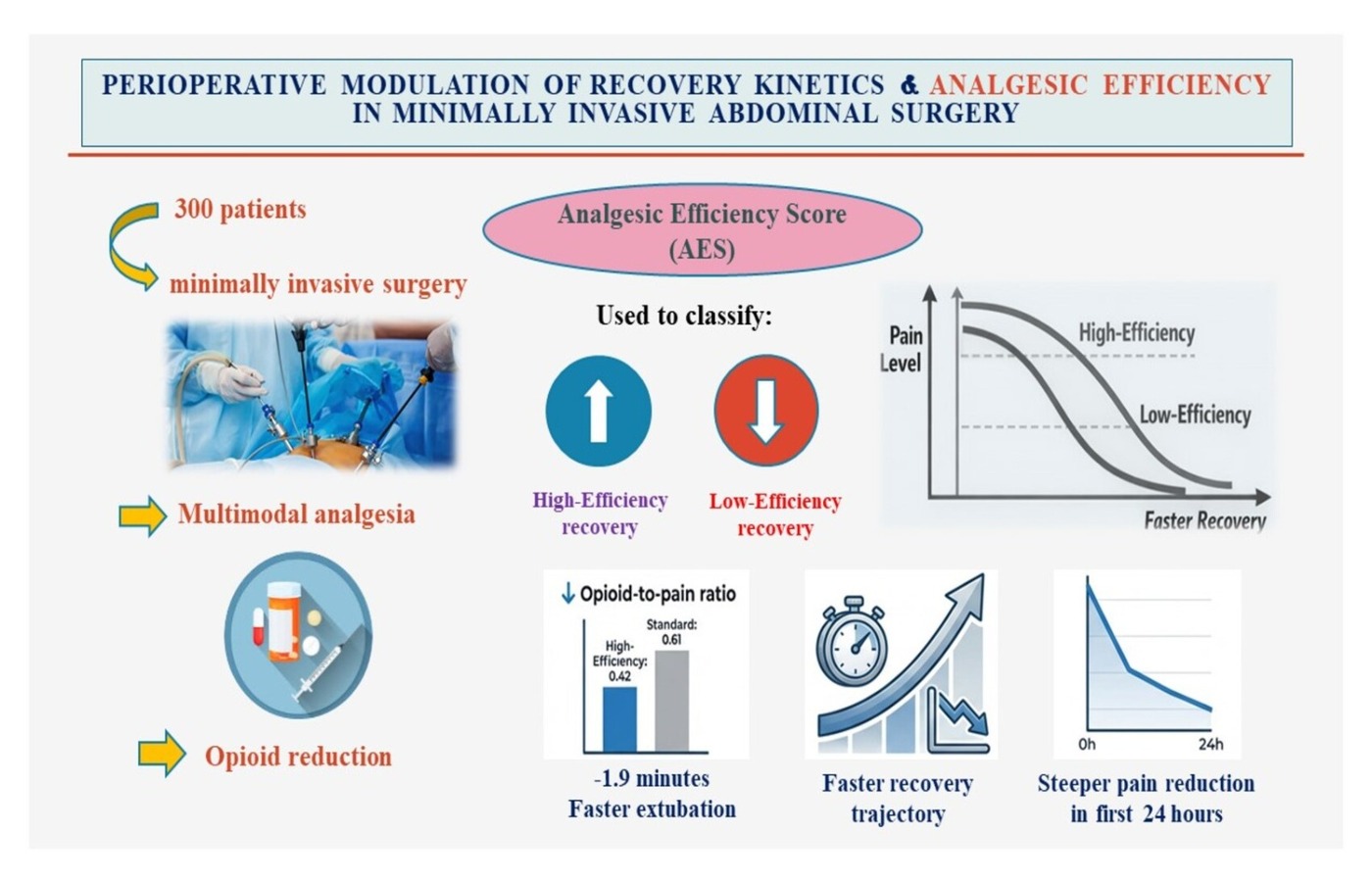
Background: The use of perioperative analgesic interventions in addition to opioid dose reduction has an impact on postoperative recovery in minimally invasive abdominal surgery. Measuring recovery dynamics and analgesic performance gives a systems perspective of postoperative time courses as opposed to drug comparison. Methods: Perioperative data on 300 adult patients, undergoing the abdominal less invasive surgeries were included in this secondary analytical study. The recovery kinetics were evaluated based on time-dependent outcomes as long as extubation latency, early postoperative pain slope and accumulated opioid exposure divided by pain burden. A combination of opioid intake and 24 hours serial pain assessment was used to create an Analgesic Efficiency Score (AES). The patients were stratified to high- and low-efficiency recovery profiles. Results: High-efficiency recovery patients had significantly lower opioid-to-pain ratios (0.42 ± 0.11 vs. 0.61 ± 0.15, p < 0.001) and followed faster early recovery kinetics, which involve fewer extubation latency differences (mean difference -1.9-minutes, p<0.001). The relationship between the intensity of pain and pain resolution after surgery in efficient recovery was steeper with a higher decrease in the level of pain during the initial 24 hours after surgery. Multivariate analysis found that the analgesic efficiency was an independent predictor of faster recovery kinetics (adjusted R2 = 0.38) regardless of the demographics or the duration of the surgery. Conclusion: Secondary recovery-oriented analysis proves that analgesic efficiency with opioid reduction is a major determinant of postoperative kinetics of recoveries in the case of minimally invasive abdominal surgery.
References
1. Hossain KZ, Kamarajan C. Advances in minimally invasive surgical techniques outcomes and challenges in complex abdominal procedures. Pac J Med Res. 2024 Dec 31;1(1):4–13. https://doi.org/10.70818/pjmr.2024.v01i01.06
2. Panditrao MM. Opioids: boon or bane? It does not matter! A clarion call for opioid sparing strategy. Adesh Univ J Med Sci Res. 2025 May 12;6(2):79–85. https://doi.org/10.25259/AUJMSR_9_2025
3. Shinn D, Mok JK, Vaishnav AS, Louie PK, Sivaganesan A, Shahi P, et al. Recovery kinetics after commonly performed minimally invasive spine surgery procedures. Spine. 2022 Nov 1;47(21):1489–1496. https://doi.org/10.1097/BRS.0000000000004399
4. Jaramillo S, Marco-Ariño N, Montane-Muntane M, Blasi A, Navarro-Ripoll R, de Peray-Bruel C, et al. Mechanism-based modeling of perioperative variations in hemoglobin concentration in patients undergoing laparoscopic surgery. Anesth Analg. 2024 Jan 1;138(1):141–151. https://doi.org/10.1213/ANE.0000000000006634
5. Cavaglià M, Zizzi EA, Dombrowski S, Deriu MA, Tuszynski JA. Alteration of consciousness by anaesthetics: a multiscale modulation from the molecular to the systems level. J Conscious Stud. 2022 Jun 1;29(5–6):21–49. https://doi.org/10.53765/20512201.29.5.021
6. Alfieri A, Di Franco S, Maffei V, Sansone P, Pace MC, Passavanti MB, et al. Phytochemical modulators of nociception: a review of cannabis terpenes in chronic pain syndromes. Pharmaceuticals. 2025 Jul 24;18(8):1100. https://doi.org/10.3390/ph18081100
7. Wu M, Park B, Chu XP. Nociceptin and the NOP receptor in pain management: from molecular insights to clinical applications. Anesth Res. 2025 Aug 11;2(3):18. https://doi.org/10.3390/anesthres2030018
8. Mithany RH, Nesma D, Hasaan SM, Samana A, Abdelmaseeh M, Farid G, et al. Revolutionizing surgical care: the power of enhanced recovery after surgery (ERAS). Cureus. 2023;15(11):e48795. https://doi.org/10.7759/cureus.48795
9. Myhre M, Jacobsen HB, Andersson S, Stubhaug A. Cognitive effects of perioperative pregabalin: secondary exploratory analysis of a randomized placebo-controlled study. Anesthesiology. 2019 Jan 1;130(1):63–71. https://doi.org/10.1097/ALN.0000000000002473
10. Sullivan KM, Dean A, Soe MM. On academics: OpenEpi: a web-based epidemiologic and statistical calculator for public health. Public Health Rep. 2009 May;124(3):471–474. https://doi.org/10.1177/003335490912400320
11. Horvath B, Kloesel B, Todd MM, Cole DJ, Prielipp RC. The evolution, current value, and future of the American Society of Anesthesiologists Physical Status Classification System. Anesthesiology. 2021 Oct 12;135(5):904–919. https://doi.org/10.1097/ALN.0000000000003947
12. Bi P. A study of integrating formative self-assessment into mobile L2 reading based on the rotated component matrix analysis through SPSS 26.0. In: 2021 Int Conf on Educ, Info Management and Service Sci (EIMSS); 2021 Jul 16; pp. 546–549. IEEE. https://doi.org/10.1109/EIMSS53851.2021.00122
13. Kumar K, Kirksey MA, Duong S, Wu CL. A review of opioid-sparing modalities in perioperative pain management: methods to decrease opioid use postoperatively. Anesth Analg. 2017 Nov 1;125(5):1749–1760. https://doi.org/10.1213/ANE.0000000000002497
14. Ghai B, Jafra A, Bhatia N, Chanana N, Bansal D, Mehta V. Opioid sparing strategies for perioperative pain management other than regional anaesthesia: a narrative review. J Anaesthesiol Clin Pharmacol. 2022 Jan 1;38(1):3–10. https://doi.org/10.4103/joacp.JOACP_362_19
15. Xie H, Wei J, Ma Z, Ge W. Predictive factors for acute postoperative pain after open radical gastrectomy for gastric cancer. Front Public Health. 2022 Jun 1;10:907222. https://doi.org/10.3389/fpubh.2022.907222
16. Zhang K, Jiao B, Sun J, Zhang X, Zhang G, Li N, Liu B, Zhou Z. Predicting high-risk factors for postoperative inadequate analgesia and adverse reactions in cesarean delivery surgery: a prospective study. Int J Surg. 2025 Jun 1;111(6):3859–3875. https://doi.org/10.1097/JS9.0000000000002354
17. Chen D, Li X, Chen Y, Zeng H, Liu J, Li Q. Opioid dose, pain, and recovery following abdominal surgery: a retrospective cohort study. J Clin Med. 2022 Dec 9;11(24):7320. https://doi.org/10.3390/jcm11247320
18. Nunes JC, Costa GP, Weleff J, Rogan M, Compton P, De Aquino JP. Assessing pain in persons with opioid use disorder: approaches, techniques and special considerations. Br J Clin Pharmacol. 2024 Dec;90(12):2985–3002. https://doi.org/10.1111/bcp.16055
19. Vasilopoulos T, Wardhan R, Rashidi P, Fillingim RB, Wallace MR, Crispen PL, et al. Patient and procedural determinants of postoperative pain trajectories. Anesthesiology. 2021 Mar 1;134(3):421–437. https://doi.org/10.1097/ALN.0000000000003681
20. Fang L, Wang Q, Xu Y. Postoperative discharge scoring criteria after outpatient anesthesia: a review of the literature. J PeriAnesth Nurs. 2023 Aug 1;38(4):642–649. https://doi.org/10.1016/j.jopan.2022.11.008
21. Boorman DC, Rehal SK, Fazili M, Martin LJ. Sex and strain differences in analgesic and hyperlocomotor effects of morphine and μ-opioid receptor expression in mice. J Neurosci Res. 2025 Apr;103(4):e70039. https://doi.org/10.1002/jnr.70039
22. Leng Y, Teng Y, Liu J, Zou X, Ou M, Zhu T, et al. The modulatory effects of anesthetics and analgesics on neurophysiological monitoring and underlying mechanisms. Curr Neuropharmacol. 2025 Feb 18. https://doi.org/10.2174/011570159X349119250127104107
23. Grant MC, Chappell D, Gan TJ, Manning MW, Miller TE, Brodt JL, et al. Pain management and opioid stewardship in adult cardiac surgery: joint consensus report of the PeriOperative Quality Initiative and the Enhanced Recovery After Surgery Cardiac Society. J Thorac Cardiovasc Surg. 2023 Dec 1;166(6):1695–1706. https://doi.org/10.1016/j.jtcvs.2023.01.020
24. Toscano A, Scimia P, Fusco P. Multimodal opioid sparing. In: Fast-Track in Cardiac Anesthesia: Cardiac Surgery in the Era of ERAS. Cham: Springer Nature Switzerland; 2024 Dec 25. p. 33–41. https://doi.org/10.1007/978-3-031-70899-2_4
25. Esteve-Pérez N, Perez-Herrero MA, Montes-Perez A, López-Alvarez S, de Dolor Agudo GD. Management of acute postoperative pain: conditions to guarantee the safety and effectiveness of analgesic treatments. Rev Esp Anestesiol Reanim (Engl Ed). 2024 Apr 1;71(4):304–316. https://doi.org/10.1016/j.redare.2022.07.005
Published
Issue
Section
License
Copyright (c) 2025 Ayesha Shafqat, Muhammad Ahmed Saleem, Muhammad Amjad Khan (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





