Regulated Cerebral Capacity: Insights into Biophysical and Metabolic Boundaries of Neural Networks-A Narrative Review
Keywords:
Brain, Cerebral Cortex, Cognition, Neuroimaging, Neuronal Plasticity, Synaptic Transmission, Neural Networks, Metabolism, Functional SpecializationAbstract
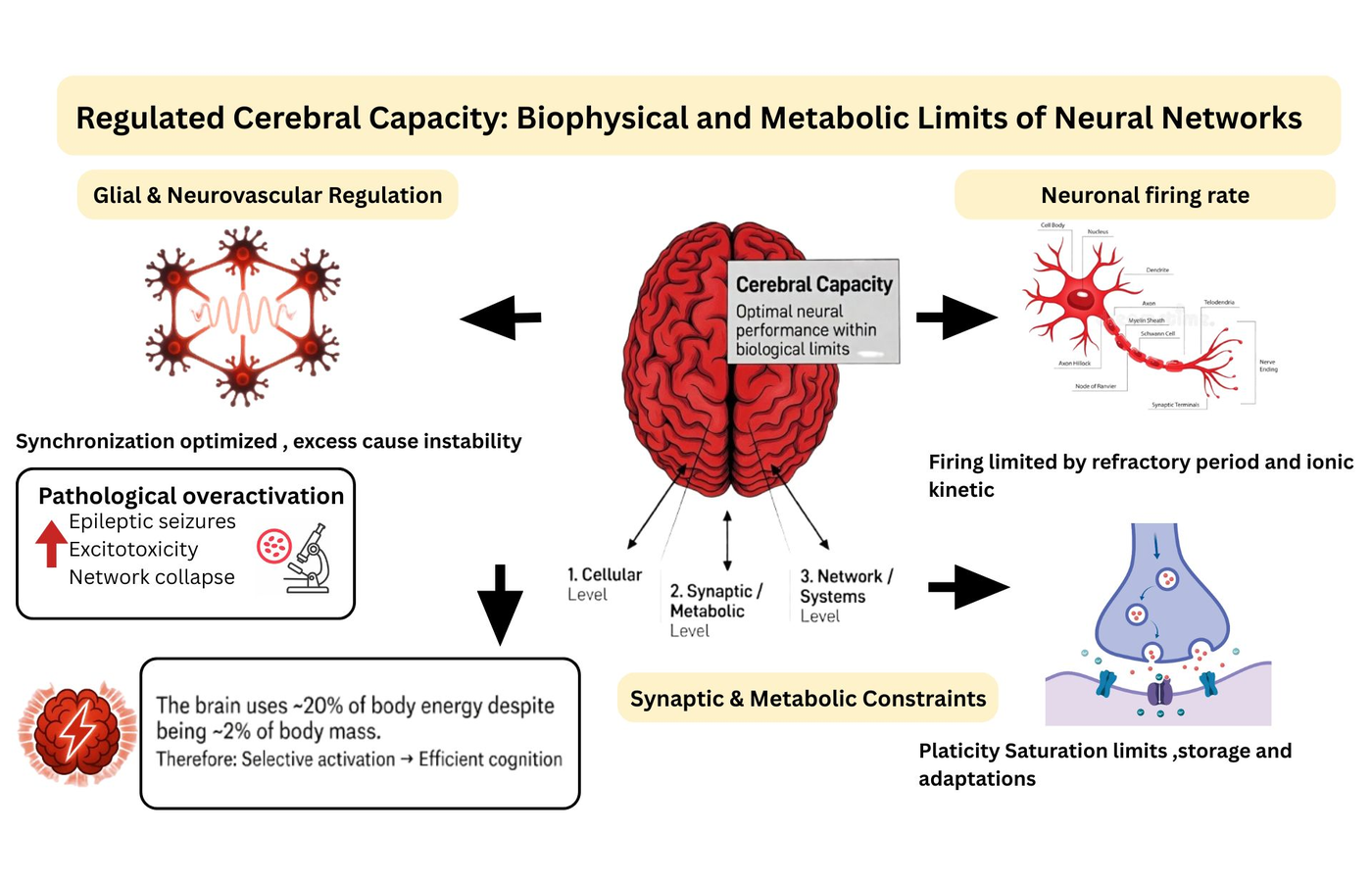
The human beings have an amazing brain capacity but it is limited at the biological, cellular and network level. Common assertions that people use only a minor part (10%) of their brains are not scientifically proven and ignore the complicated control that governs brain activity. Cerebral capacity is the optimal performance of neural systems under physiological conditions, which does not follow any theoretical activation limit. At cellular level, ion channels and refractory periods limit neuronal excitation, whereas receptor concentrations, plasticity exhaustion, and neurotransmitter cycling all limit synaptic communication. On network level, synchronized coordination is required to process information but it is intrinsically restricted to avoid instability and dysfunction. The functional specialization includes selective recruitment of different brain parts according to task demands rather than maximum usage to meet some hypothetical maximized level. Neuroimaging research tends to misinterpret task specific activations as the only functional regions of brain, whereas background neural activity is omnipresent to ensure homeostasis, preparedness, and metabolic stability. The unused brain myth disregards the evolutionary and energetic needs of preservation of metabolically active and functionally necessary structures. This review combines molecular, cellular, and systems neuroscience to explain how cerebral capacity is naturally controlled. By providing historical, conceptual and mechanistic perspectives, it suggests that neural activity does not work for maximum functionality, instead stability, efficiency and adaptability of the system is ensured through biological regulation. Moreover, it discredits the brain capacity related myths and offers fundamental understanding about constraints of human brain capacity, from neurons to large-scale networks.
References
1. Li T, Zheng Y, Wang Z, Zhu DC, Ren J, Liu T, et al. Brain information processing capacity modeling. Sci Rep. 2022;12(1):2174. https://doi.org/10.1038/s41598-022-07499-4
2. Papo D, Buldú JM. A complex network perspective on brain disease. Biol Rev. 2026;101(1):364-99. https://doi.org/10.1111/brv.70086
3. Hengen KB, Shew WL. Is criticality a unified setpoint of brain function? Neuron. 2025;113(16):2582-98. https://doi.org/10.1016/j.neuron.2025.05.020
4. Liu Y, Shen X, Zhang Y, Zheng X, Cepeda C, Wang Y, et al. Interactions of glial cells with neuronal synapses, from astrocytes to microglia and oligodendrocyte lineage cells. Glia. 2023;71(6):1383-401. https://doi.org/10.1002/glia.24343
5. Seguin C, Sporns O, Zalesky A. Brain network communication: concepts, models and applications. Nat Rev Neurosci. 2023;24(9):557-74. https://doi.org/10.1038/s41583-023-00718-5
6. Swanson LW, Hahn JD, Sporns O. Neural network architecture of a mammalian brain. Proc Natl Acad Sci U S A. 2024;121(39):e2413422121. https://doi.org/10.1073/pnas.2413422121
7. Yen C, Lin CL, Chiang MC. Exploring the frontiers of neuroimaging: a review of recent advances in understanding brain functioning and disorders. Life. 2023;13(7):1472. https://doi.org/10.3390/life13071472
8. Perich MG, Narain D, Gallego JA. A neural manifold view of the brain. Nat Neurosci. 2025;28(8):1582-97. https://doi.org/10.1038/s41593-025-02031-z
9. Anum SR, Shafiq M, Fatima S, Bibi A, Khan AU, Naz S, et al. Evolution of the human brain and the myth of its ten-percent use. Soc Evol Hist. 2024;23(2):44-69. https://doi.org/10.30884/seh/2024.02.02
10. Vaidya AR, Pujara MS, Petrides M, Murray EA, Fellows LK. Lesion studies in contemporary neuroscience. Trends Cogn Sci. 2019;23(8):653-71. https://doi.org/10.1016/j.tics.2019.05.009
11. Heald JB, Wolpert DM, Lengyel M. The computational and neural bases of context-dependent learning. Annu Rev Neurosci. 2023;46:233-58. https://doi.org/10.1146/annurev-neuro-092322-100402
12. Bolaños JP, Magistretti PJ. The neuron–astrocyte metabolic unit as a cornerstone of brain energy metabolism in health and disease. Nat Metab. 2025;7:1-10. https://doi.org/10.1038/s42255-025-01404-9
13. Solms M. “Function” in functional neurological disorders: the common ground of neuroscience and psychoanalysis. Neuropsychoanalysis. 2025;27:1-4. https://doi.org/10.1080/15294145.2025.2472340
14. Drukarch B, Wilhelmus MM. Thinking about the action potential: the nerve signal as a window to the physical principles guiding neuronal excitability. Front Cell Neurosci. 2023;17:1232020. https://doi.org/10.3389/fncel.2023.1232020
15. Puttkammer F, Lindner B. Fluctuation–response relations for integrate-and-fire models with an absolute refractory period. Biol Cybern. 2024;118(1):7-19. https://doi.org/10.1007/s00422-023-00982-9
16. Steinhardt CR, Mitchell DE, Cullen KE, Fridman GY. Pulsatile electrical stimulation creates predictable, correctable disruptions in neural firing. Nat Commun. 2024;15(1):5861. https://doi.org/10.1038/s41467-024-49900-y
17. Fabbri R, Botte E, Ahluwalia A, Magliaro C. Digitoids: a novel computational platform for mimicking oxygen-dependent firing of neurons in vitro. Front Neuroinform. 2025;19:1549916. https://doi.org/10.3389/fninf.2025.1549916
18. West AE. Genetic mechanisms of experience-dependent neuronal plasticity. Annu Rev Genet. 2025;59:1-22. https://doi.org/10.1146/annurev-genet-020325-103824
19. Hagena H, Manahan-Vaughan D. Interplay of hippocampal long-term potentiation and long-term depression in enabling memory representations. Philos Trans R Soc Lond B Biol Sci. 2024;379(1906):20230229. https://doi.org/10.1098/rstb.2023.0229
20. Incontro S, Musella ML, Sammari M, Di Scala C, Fantini J, Debanne D. Lipids shape brain function through ion channel and receptor modulations: physiological mechanisms and clinical perspectives. Physiol Rev. 2025;105(1):137-207. https://doi.org/10.1152/physrev.00004.2024
21. Papatheodoropoulos C. Compensatory regulation of excitation/inhibition balance in the ventral hippocampus: insights from fragile X syndrome. Biology. 2025;14(4):363. https://doi.org/10.3390/biology14040363
22. Dufor T, Lohof AM, Sherrard RM. Magnetic stimulation as a therapeutic approach for brain modulation and repair: underlying molecular and cellular mechanisms. Int J Mol Sci. 2023;24(22):16456. https://doi.org/10.3390/ijms242216456
23. Amaral-Silva L, Santin JM. Synaptic modifications transform neural networks to function without oxygen. BMC Biol. 2023;21(1):54. https://doi.org/10.1186/s12915-023-01518-0
24. Singha SP, Memon S, Kazi SAF, Nizamani GS. Gamma aminobutyric acid signaling disturbances and altered astrocytic morphology associated with Bisphenol A induced cognitive impairments in rat offspring. Birth Defects Res. 2021;113(12):911-924. https://doi.org/10.1002/bdr2.1886
25. Singha SP, Memon S, Bano U, Isaac AD, Shahani MY. Evaluation of p21 expression and related autism-like behavior in Bisphenol-A exposed offspring of Wistar albino rats. Birth Defects Res. 2022;114(11):536-550. https://doi.org/10.1002/bdr2.2022
26. Li S, Sheng ZH. Energy matters: presynaptic metabolism and the maintenance of synaptic transmission. Nat Rev Neurosci. 2022;23(1):4-22. https://doi.org/10.1038/s41583-021-00535-8
27. Alabdali A, Ben Bacha A, Alonazi M, Al-Ayadhi LY, Alanazi AS, El-Ansary A. Comparative evaluation of certain biomarkers emphasizing abnormal GABA inhibitory effect and glutamate excitotoxicity in autism spectrum disorders. Front Psychiatry. 2025;16:1562631. https://doi.org/10.3389/fpsyt.2025.1562631
28. van Susteren H, Rose CR, van Putten MJ, Meijer HG. The role of the glutamate–glutamine cycle in synaptic transmission during ischemia and recovery. bioRxiv. 2025. https://doi.org/10.1101/2025.11.10.687543
29. Li H, Zhao Y, Dai R, Geng P, Weng D, Wu W, et al. Astrocytes release ATP/ADP and glutamate in flashes via vesicular exocytosis. Mol Psychiatry. 2025;30(6):2475-89. https://doi.org/10.1038/s41380-024-02851-8
30. Wawrzyniak A, Krawczyk-Marć I, Żuryń A, Walocha J, Balawender K. Diversity, functional complexity, and translational potential of glial cells in the central nervous system. Int J Mol Sci. 2025;26(18):9080. https://doi.org/10.3390/ijms26189080
31. Larios A, Sullere S, Gu C. Cerebral blood flow and energy demand: imaging insights into neurovascular function. Neurophotonics. 2025;12(S2):S22810. https://doi.org/10.1117/1.NPh.12.S2.S22810
32. Coccarelli A, Polydoros I, Drysdale A, Harraz OF, Kadapa C. A computational framework for quantifying blood flow dynamics across myogenically-active cerebral arterial networks. Biomech Model Mechanobiol. 2025;24:1-8. https://doi.org/10.1007/s10237-025-01958-3
33. Joseph UG, Oyovwi MO, Jeroh E, Esuku DT, Ben-Azu B. Dysfunctional astrocyte metabolism: a driver of imbalanced excitatory/inhibitory tone and support for therapeutic intervention targets. J Mol Pathol. 2025;6(2):12. https://doi.org/10.3390/jmp6020012
34. Affrald RJ, Narayan S. Oligodendrocytes in neuronal axonal conduction and methods for enhancing their performance. Int J Neurosci. 2025;135(12):1328-49. https://doi.org/10.1080/00207454.2024.2362200
35. Mao R, Zong N, Hu Y, Chen Y, Xu Y. Neuronal death mechanisms and therapeutic strategy in ischemic stroke. Neurosci Bull. 2022;38(10):1229-47. https://doi.org/10.1007/s12264-022-00859-0
36. Tort AB, Laplagne DA, Draguhn A, Gonzalez J. Global coordination of brain activity by the breathing cycle. Nat Rev Neurosci. 2025;26:1-21. https://doi.org/10.1038/s41583-025-00920-7
37. Hao Y, Tower T, Lax H, Hütt MT, Graham DJ. Brain volume predicts survival of colliding-spreading messages on mammal brain networks. arXiv. 2025. https://doi.org/10.48550/arXiv.2505.15477
38. Voicu V, Toader C, Șerban M, Covache-Busuioc RA, Ciurea AV. Systemic neurodegeneration and brain aging: multi-omics disintegration, proteostatic collapse, and network failure across the CNS. Biomedicines. 2025;13(8):2025. https://doi.org/10.3390/biomedicines13082025
39. Keitel A, Keitel C, Alavash M, Bakardjian K, Benwell CS, Bouton S, et al. Brain rhythms in cognition—controversies and future directions. arXiv. 2025. https://doi.org/10.48550/arXiv.2507.15639
40. Cuesta P, Ochoa-Urrea M, Funke M, Hasan O, Zhu P, Marcos A, et al. Gamma band functional connectivity reduction in patients with amnestic mild cognitive impairment and epileptiform activity. Brain Commun. 2022;4(2):fcac012. https://doi.org/10.1093/braincomms/fcac012
41. Liang J, Wang SJ, Zhou C. Less is more: wiring-economical modular networks support self-sustained firing-economical neural avalanches for efficient processing. Natl Sci Rev. 2022;9(3):nwab102. https://doi.org/10.1093/nsr/nwab102
42. Shaikh P, Bano U, Memon S, Bano S, Gul S, Pirya. Neuroanatomical study of in-vivo brainstem abnormalities in autism spectrum disorder and their clinical correlations. Pak J Med Dent. 2025;14(3). https://doi.org/10.36283/ziun-pjmd14-3/039
43. Ghafoor A, Kashif S, Ali SMS, Memon S, Memon SS, Mehmood A, et al. Evaluating neurotherapeutic potential of naringenin by true experiments: insights into in-vivo psychiatry care models. Pak J Med Dent. 2025;14(3). https://doi.org/10.36283/ziun-pjmd14-3/043
44. Lu J, Xu F, Lu X, Wang T, Huang S, Zhang J. Synchronous firing transition between different regions of brain neural networks enhanced by Hamiltonian energy. Nonlinear Dyn. 2025;113(10):12143-50. https://doi.org/10.1007/s11071-024-10676-x
45. Andrew RD, Farkas E, Hartings JA, Brennan KC, Herreras O, Müller M, et al. Questioning glutamate excitotoxicity in acute brain damage: the importance of spreading depolarization. Neurocrit Care. 2022;37(Suppl 1):11-30. https://doi.org/10.1007/s12028-021-01429-4
46. Masoli S, Rizza MF, Tognolina M, Prestori F, D’Angelo E. Computational models of neurotransmission at cerebellar synapses unveil the impact on network computation. Front Comput Neurosci. 2022;16:1006989. https://doi.org/10.3389/fncom.2022.1006989
47. Calabrese RL, Marder E. Degenerate neuronal and circuit mechanisms important for generating rhythmic motor patterns. Physiol Rev. 2025;105(1):95-135. https://doi.org/10.1152/physrev.00003.2024
48. Shen G, Zhao D, Dong Y, Zeng Y. Brain-inspired neural circuit evolution for spiking neural networks. Proc Natl Acad Sci U S A. 2023;120(39):e2218173120. https://doi.org/10.1073/pnas.2218173120
49. Friedman R. A perspective on information optimality in a neural circuit and other biological systems. Signals. 2022;3(2):410-27. https://doi.org/10.3390/signals3020025
50. Bano U, Memon S, Shahani MY, Shaikh P, Gul S. Epigenetic effects of in utero bisphenol A administration: diabetogenic and atherogenic changes in mice offspring. Iran J Basic Med Sci. 2019;22(5):521-528. https://doi.org/10.22038/ijbms.2019.29909.7357
51. Rae CD, Baur JA, Borges K, Dienel G, Díaz-García CM, Douglass SR, et al. Brain energy metabolism: a roadmap for future research. J Neurochem. 2024;168(5):910-54. https://doi.org/10.1111/jnc.16032
52. Hunter P. Understanding redundancy and resilience: redundancy in life is provided by distributing functions across networks rather than back-up systems. EMBO Rep. 2022;23(3):e54742. https://doi.org/10.15252/embr.202254742
53. Duong-Tran D, Kaufmann R, Chen J, Wang X, Garai S, Xu FH, et al. Homological landscape of human brain functional sub-circuits. Mathematics. 2024;12(3):455. https://doi.org/10.3390/math12030455
54. Khanjanianpak M, Valizadeh A. Optimizing information capacity in modular neural networks through excitatory and inhibitory connectivity. Neurocomputing. 2025;130313. https://doi.org/10.1016/j.neucom.2025.130313
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Naveera Mazhar, Maha Aslam khan, Arslan Majeed, Kainat Zafar (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





