Laboratory Workflow Determinants Affecting Glucose Integrity: A Secondary Analysis of Pre-Analytical Errors
Keywords:
Pre-analytical Errors, Glucose Estimation, Laboratory Diagnosis, In-Vitro Glycolysis, Quality Control, Diagnostic PathogenesisAbstract
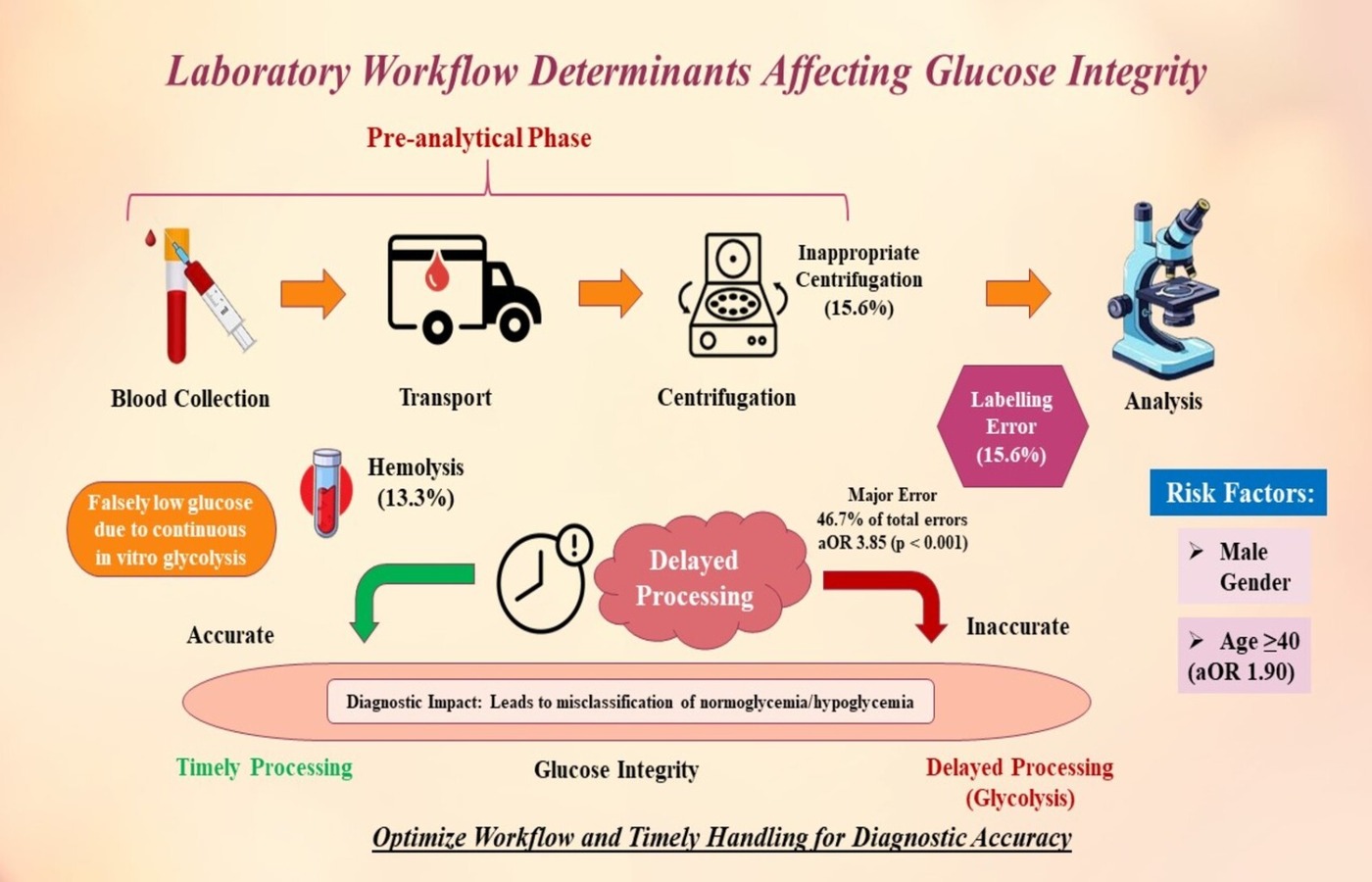
Background: Pre-analytical errors are the most critical step to the laboratory diagnostics process. The aim of the secondary analysis was to assess the distribution and predictors of the pre-analytical errors of glucose specimen collection as well as the statistical relationships between pre-analytical errors and delayed processing. Methods The secondary statistical analysis was done on 225 samples (venous blood) collected to test glucose in the Pathology Department of Bahawal Victoria Hospital, Pakistan. Pre-analytical errors, which were measured, were; delay of processing of the specimen (more than 1 hour), hemolysis, inadequate specimen quantity, improper centrifugation and labeling errors. The statistical significance was established to be p < 0.05. Results The age of the study population was 40.7 with 10.9 standard deviation with the females constituting 60 percent of the cohort. The most common error was delayed specimen processing, detected in 9.3% (n= 21) of all samples and 46.7% of the errors. The results of the logistic regression analysis revealed age 40 years and above to be an independent predictive factor of the occurrence of pre-analytical error (Adjusted OR = 1.9: 95%CI = 1.1 3.3: p = .026). The strongest correlation with the total burden of errors was found with delayed specimen processing (p < 0.001). Conclusion: This secondary analysis proves that the pre-analytical errors of glucose testing largely depend on the age of patients and delay in handling the specimen. Late processing proves to be the most significant and adjustable factor causing diagnostic error by an increased level of in vitro glycolysis.
References
1. Tasneem A, Zubair M, Rasool Z, Tareen FZ. Frequency and types of pre-analytical errors in a clinical laboratory of a specialized healthcare hospital. Pak J Med Sci. 2024 Jan;40(2 ICON Suppl):S70–S74. https://doi.org/10.12669/pjms.40.2(ICON).8963
2. Nordin N, Ab Rahim SN, Wan Omar WFA, Zulkarnain S, Sinha S, Kumar S, et al. Preanalytical errors in clinical laboratory testing at a glance: source and control measures. Cureus. 2024 Mar 30;16(3):e57243. https://doi.org/10.7759/cureus.57243
3. John GK, Favaloro EJ, Austin S, Islam MZ, Santhakumar AB. From errors to excellence: the pre-analytical journey to improved quality in diagnostics. A scoping review. Clin Chem Lab Med. 2025 Jan 28;63(7):1243–1259. https://doi.org/10.1515/cclm-2024-1277
4. Al Sula KM, Almalki AM, Alzaidi HM, Alshanbar TA, Althobiti BH, Alharb YA, et al. A comprehensive review of errors in medical laboratories and strategies for improvement. J Int Crisis Risk Commun Res. 2024;7(Suppl 9):1902. https://doi.org/10.63278/jicrcr.vi.917
5. Lin Y, Spies NC, Zohner K, McCoy D, Zaydman MA, Farnsworth CW. Pre-analytical phase errors constitute the vast majority of errors in clinical laboratory testing. Clin Chem Lab Med. 2025 May 5;63(9):1709–1715. https://doi.org/10.1515/cclm-2025-0190
6. Alcantara JC, Alharbi B, Almotairi Y, Alam MJ, Muddathir ARM, Alshaghdali K. Analysis of preanalytical errors in a clinical chemistry laboratory: a 2-year study. Medicine (Baltimore). 2022 Jul 8;101(27):e29853. https://doi.org/10.1097/MD.0000000000029853
7. Li B, Cai X, Zhan L, Zhang X, Lin Y, Zeng J. Quality control circle practices to reduce specimen rejection rates. J Multidiscip Healthc. 2024 Oct 26;17:4925–4935. https://doi.org/10.2147/JMDH.S486276
8. Sacks DB, Arnold M, Bakris GL, Bruns DE, Horvath AR, Lernmark Å, et al. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Diabetes Care. 2023 Oct 1;46(10):e151–e199. https://doi.org/10.2337/dci23-0036
9. Coelho H, Silva F, Correia M, Rodrigues PM. Artificial intelligence in patient blood management: a systematic review of predictive, diagnostic, and decision support applications. J Clin Med. 2025 Nov 29;14(23):8479. https://doi.org/10.3390/jcm14238479
10. Güner Y, Güner EK, Üçüncüoğlu M, Yüksel H. Evaluation of specimen rejection rates in the preanalytical phase and nurses’ experiences: a mixed design study. BMC Nurs. 2025 Jul 1;24(1):705. https://doi.org/10.1186/s12912-025-03426-w
11. Bhardwaj R, Agrawal U, Vashist P, Manna S. Determination of sample size for various study designs in medical research: a practical primer. J Fam Med Prim Care. 2024 Jul;13(7):2555–2561. https://doi.org/10.4103/jfmpc.jfmpc_1675_23
12. Azocar González I, González-González ML, Sepúlveda Maturana F, Azocar González C, Ramírez-Pereira M. Pre-analytical errors in clinical laboratories: an integrative review. Enfermería Cuidados Humanizados. 2024 Dec;13(2):e21214. https://doi.org/10.22235/ech.v13i2.4223
13. Dundar C, Bahadir O. Preanalytical errors in clinical biochemistry laboratory and relationship with hospital departments and staff: a record-based study. J Patient Saf. 2023 Jun 1;19(4):239–242. https://doi.org/10.1097/PTS.0000000000001115
14. Piskunov DP, Danilova LA, Pushkin AS, Rukavishnikova SA. Influence of exogenous and endogenous factors on the quality of the preanalytical stage of laboratory tests (review of literature). Klin Lab Diagn. 2020 Dec 29;65(12):778–784. https://doi.org/10.18821/0869-2084-2020-65-12-778-784
15. Ialongo C. Blood alcohol concentration in the clinical laboratory: a narrative review of the preanalytical phase in diagnostic and forensic testing. Biochem Med (Zagreb). 2024 Feb 15;34(1):010501. https://doi.org/10.11613/BM.2024.010501
16. Zorbozan N, Zorbozan O. Evaluation of preanalytical and postanalytical phases in clinical biochemistry laboratory according to IFCC laboratory errors and patient safety specifications. Biochem Med (Zagreb). 2022 Oct 1;32(3):030701. https://doi.org/10.11613/BM.2022.030701
17. Orhan B, Sonmez D, Cubukcu HC, Zengi O, Ozturk Emre H, Cinaroglu I, et al. The use of preanalytical quality indicators: a Turkish preliminary survey study. Clin Chem Lab Med. 2020 Oct 20;59(5):837–843. https://doi.org/10.1515/cclm-2020-1426
18. Giavarina D. The pre-analytical phase. In: Clinical and Laboratory Medicine. Cham: Springer International Publishing; 2024 Feb 22. p. 47–56. https://doi.org/10.1007/978-3-031-24958-7_6
19. Gaikwad K, Selkar S. Evaluation of pre-analytical errors in clinical biochemistry laboratory of a tertiary care center in India. J Clin Basic Res. 2021 Dec 10;5(4):1–8. https://doi.org/10.61186/jcbr.5.4.1
20. Alenezy NT, Arain SA, Omair A, Afnan R, Nadheef A, Ahmed AF, et al. Frequencies and causes of pre-analytical errors in a tertiary care hospital laboratory in Saudi Arabia. Ann Saudi Med. 2025 Nov–Dec;45(6):381–387. https://doi.org/10.5144/0256-4947.2025.381
21. Thachil A, Wang L, Mandal R, Wishart D, Blydt-Hansen T. An overview of pre-analytical factors impacting metabolomics analyses of blood samples. Metabolites. 2024 Aug 28;14(9):474. https://doi.org/10.3390/metabo14090474
22. Guo Y, Dai W, Jiang Y, Liu X. Analysis and actions after laboratory errors in a Chinese university hospital. BMC Health Serv Res. 2025 Oct 3;25(1):1296. https://doi.org/10.1186/s12913-025-13320-5
23. Kazmierczak SC, Morosyuk S, Rajkumar R. Evaluation of preanalytical point-of-care testing errors and their impact on productivity in the emergency department in the United States. J Appl Lab Med. 2022 May 4;7(3):650–660. https://doi.org/10.1093/jalm/jfab158
24. Lee L, Rankin W. Preparation for blood tests: what can go wrong before the sample reaches the lab. Aust Prescr. 2025 Aug;48(4):122–127. https://doi.org/10.18773/austprescr.2025.034
25. Hedayati M, Razavi SA, Boroomand S, Kheradmand Kia S. The impact of pre-analytical variations on biochemical analytes stability: a systematic review. J Clin Lab Anal. 2020 Dec;34(12):e23551. https://doi.org/10.1002/jcla.23551
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Asma Hafiz, Ammara Khan, Ehsan Sattar, M Irshad (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





