Molecular Inflammatory, Oxidative Stress, and Angiogenic Pathways in Diabetic Foot Ulcer Pathogenesis: A Systematic Review and Meta-Analysis
Keywords:
Diabetic Foot, Diabetes Mellitus, Wound Healing, Inflammation, Oxidative Stress, Angiogenesis, Tumor Necrosis Factor-alpha, Vascular Endothelial Growth Factor, MalondialdehydeAbstract
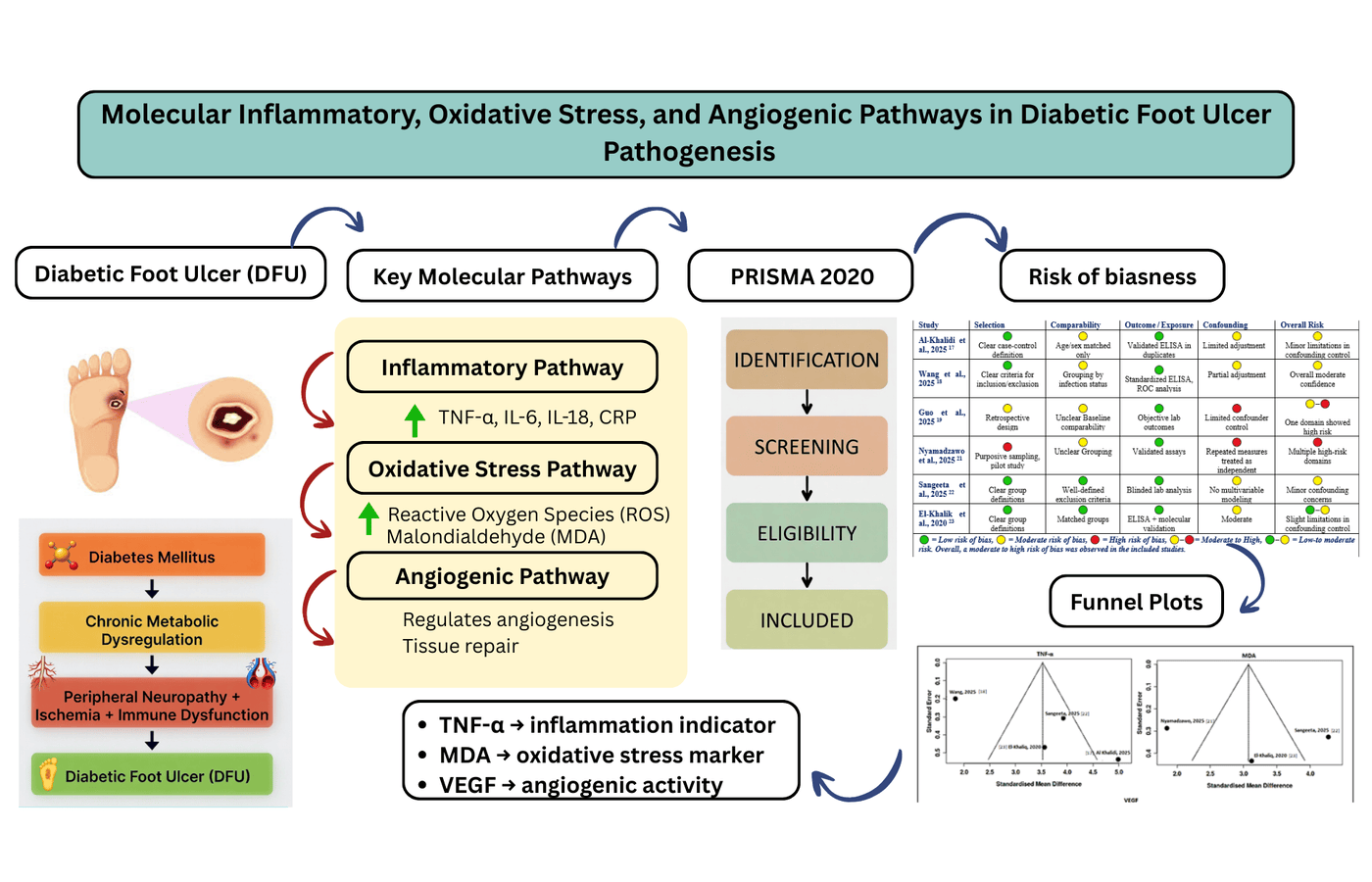
Background: Diabetic foot ulcer (DFU) is a serious health condition in diabetes mellitus and a primary reason of non-traumatic amputations of lower-limb worldwide. This study aimed to investigate the combined effect of inflammation, oxidative stress, and pathological angiogenic pathways in DFU. Methods: The extensive literature search included the use of PubMed, Web of Science, Google Scholar, and Scopus, from 2019 to 2025. Researches based on human subjects with DFUs were incorporated when they have quantitative data of inflammatory, oxidative stress or angiogenic biomarkers. Studies conducted in animal, or in vitro studies, and those without quantitative data were excluded. Funnel plot symmetry and Newcastle-Ottowa scale (NOS-adapted) was used to determine publication bias and risk of bias respectively. Results: Total seven studies fulfilled the inclusion criteria and meta-analyses were done in terms of a random-effects inverse-variance model. DFU pooled analyses showed significantly higher tumor necrosis factor-alpha (TNF-α) (SMD = 3.52; 95% CI: 2.22-4.83, p = 0.0001) and malondialdehyde (MDA) (SMD = 3.07; 95% CI: 1.67-4.48, p = 0.0001) levels in DFU when compared with controls whereas, the pooled analyses of vascular endothelial growth factor (VEGF) suggested similar pattern between the compared groups (SMD = 0.39; 95% CI: - 3.41 to 4.19, p = 0.84). Substantial heterogeneity was found (I2 > 90%). There was moderate risk of bias among included studies and certainty of evidence was also moderate according to GRADE assessment. Conclusion: High TNF-α and MDA concentrations could be utilized as molecular predictors for risk stratification by DFU and disease severity.
References
1. Wang X, Yuan CX, Xu B, Yu Z. Diabetic foot ulcers: classification, risk factors and management. World J Diabetes. 2022;13(12):1049. https://doi.org/10.4239/wjd.v13.i12.1049
2. Akkus G, Sert M. Diabetic foot ulcers: a devastating complication of diabetes mellitus continues non-stop in spite of new medical treatment modalities. World J Diabetes. 2022;13(12):1106. https://doi.org/10.4239/wjd.v13.i12.1106
3. Gong H, Ren Y, Li Z, Zha P, Bista R, Li Y, et al. Clinical characteristics and risk factors of lower extremity amputation in diabetic inpatients with foot ulcers. Front Endocrinol (Lausanne). 2023;14:1144806. https://doi.org/10.3389/fendo.2023.1144806.
4. Huang X, Li Z, Zhai Z, Wang Q, Wei R, Mo J, et al. Association between metabolic dysfunction-associated fatty liver disease and MACCEs in patients with diabetic foot ulcers: an ambispective longitudinal cohort study. Diabetes Metab Syndr Obes. 2024;17:1119–1130. https://doi.org/10.2147/DMSO.S447897
5. Deng H, Li B, Shen Q, Zhang C, Kuang L, Chen R, et al. Mechanisms of diabetic foot ulceration: a review. J Diabetes. 2023;15(4):299–312. https://doi.org/10.1111/1753-0407.13372
6. Mykhaylichenko V, Kaibov I, Bondarenko N, Parshin D, Puchkina G. Characteristics of immunoreactivity in patients with various forms of diabetic foot ulcer. Archiv Euromedica. 2022;12(3):17. https://doi.org/10.35630/2199-885X/2022/12/3.17
7. González P, Lozano P, Ros G, Solano F. Hyperglycemia and oxidative stress: an integral, updated and critical overview of their metabolic interconnections. Int J Mol Sci. 2023;24(11):9352. https://doi.org/10.3390/ijms24119352
8. Xiong Y, Chu X, Yu T, Knoedler S, Schroeter A, Lu L, et al. Reactive oxygen species-scavenging nanosystems in the treatment of diabetic wounds. Adv Healthc Mater. 2023;12(25):2300779. https://doi.org/10.1002/adhm.202300779
9. Rai V, Moellmer R, Agrawal DK. Stem cells and angiogenesis: implications and limitations in enhancing chronic diabetic foot ulcer healing. Cells. 2022;11(15):2287. https://doi.org/10.3390/cells11152287
10. Goswami AG, Basu S, Huda F, Pant J, Ghosh Kar A, Banerjee T, et al. An appraisal of vascular endothelial growth factor (VEGF): the dynamic molecule of wound healing and its current clinical applications. Growth Factors. 2022;40(3–4):73–88. https://doi.org/10.1080/08977194.2022.2074843
11. Xu J, Gao J, Li H, Zhu Z, Liu J, Gao C. The risk factors in diabetic foot ulcers and predictive value of prognosis of wound tissue vascular endothelium growth factor. Scientific Reports. 2024;14(1):14120. https://doi.org/10.1038/s41598-024-64009-4
12. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71
13. Fekete JT, Győrffy B. MetaAnalysisOnline.com: web-based tool for the rapid meta-analysis of clinical and epidemiological studies. J Med Internet Res. 2025;27:e64016. https://doi.org/10.2196/64016
14. Carra MC, Romandini P, Romandini M. Risk of bias evaluation of cross-sectional studies: adaptation of the Newcastle–Ottawa Scale. J Periodontal Res. 2025. https://doi.org/10.1111/jre.13405
15. Shao SC, Kuo LT, Huang YT, Lai PC, Chi CC. Using Grading of Recommendations Assessment, Development, and Evaluation (GRADE) to rate the certainty of evidence of study outcomes from systematic reviews: a quick tutorial. Dermatol Sin. 2023;41(1):3–7. https://doi.org/10.4103/ds.DS-D-22-00154
16. Maier M, VanderWeele TJ, Mathur MB. Using selection models to assess sensitivity to publication bias: a tutorial and call for more routine use. Campbell Syst Rev. 2022;18(3):e1256. https://doi.org/10.1002/cl2.1256
17. Al-Khalidi AAK, Hatem RM. Assessment of immunological biomarkers in diabetic patients with and without foot ulcers. Int J Basic Appl Sci. 2025;14(7):88–96. https://doi.org/10.14419/3dypmj38
18. Wang S, Gao L, Qin X, Li T, Wang J, Xie H. The prognostic and diagnostic significance of inflammatory markers TNF-α, IL-6, and IFN-γ in evaluating disease severity in diabetic foot infection. Front Cell Infect Microbiol. 2025;15:1606612. https://doi.org/10.3389/fcimb.2025.1606612
19. Guo H, Xue Z, Mei S, Li T, Yu H, Ning T, et al. Clinical efficacy of antibiotic-loaded bone cement and negative pressure wound therapy in multidrug-resistant organism diabetic foot ulcers: a retrospective analysis. Front Cell Infect Microbiol. 2025;14:1521199. https://doi.org/10.3389/fcimb.2024.1521199
20. Dhamodharan U, Karan A, Sireesh D, Vaishnavi A, Somasundar A, Rajesh K, et al. Tissue-specific role of Nrf2 in the treatment of diabetic foot ulcers during hyperbaric oxygen therapy. Free Radic Biol Med. 2019;138:53–62. https://doi.org/10.1016/j.freeradbiomed.2019.04.031
21. Nyamadzawo AT, Nishio J, Ogawa T, Okada S. Relationship between oxidative stress and severity of diabetic foot ulcers among patients with type 2 diabetes mellitus in Japan: a cross-sectional study. Health Sci Rep. 2025;8(7):e70935. https://doi.org/10.1002/hsr2.70935
22. Sangeeta S, Siripuram C, Konka S, Vaithilingam K, Periasamy P, Velu RK, et al. Biomarkers of inflammation, oxidative stress, and endothelial dysfunction in early detection of diabetic foot ulcers. Cureus. 2025;17(4):e82174. https://doi.org/10.7759/cureus.82174
23. Abd El-Khalik SR, Hafez YM, Elkholy RA. The role of circulating soluble fms-like tyrosine kinase-1 in patients with diabetic foot ulcer: a possible mechanism of pathogenesis via a novel link between oxidative stress, inflammation and angiogenesis. Microvasc Res. 2020;130:103987. https://doi.org/10.1016/j.mvr.2020.103987
24. Rashad N, Khalil U, Elshamy A, Nawara AM. Diagnostic and prognostic significance of tumor necrosis factor-α serum and mRNA expression in patients with infected diabetic foot ulcers. Zagazig Univ Med J. 2024;30(1):65–75. https://doi.org/10.21608/zumj.2023.226201.2835
25. Musale V, Wasserman DH, Kang L. Extracellular matrix remodelling in obesity and metabolic disorders. Life Metab. 2023;2(4):load021. https://doi.org/10.1093/lifemeta/load021
26. Worsley AL, Lui DH, Ntow-Boahene W, Song W, Good L, Tsui J. The importance of inflammation control for the treatment of chronic diabetic wounds. Int Wound J. 2023;20(6):2346–2359. https://doi.org/10.1111/iwj.14048
27. Deng L, Wang G, Ju S. Correlation between inflammatory factors, autophagy protein levels, and infection in granulation tissue of diabetic foot ulcer. Immun Inflamm Dis. 2024;12(4):e1233. https://doi.org/10.1002/iid3.1233
28. Memon SR, Brohi GH, Memon FR, Shahani MY, Memon S. Study on red cell distribution width, haematocrit and red blood corpuscle (RBC) indices as early markers for the detection of coronary artery disease: a case control study. Prof Med J. 2019;26(12):2075–2079. https://doi.org/10.29309/TPMJ/2019.26.12.3069
29. Dri E, Lampas E, Lazaros G, Lazarou E, Theofilis P, Tsioufis C, et al. Inflammatory mediators of endothelial dysfunction. Life. 2023;13(6):1420. https://doi.org/10.3390/life13061420
30. Nirenjen S, Narayanan J, Tamilanban T, Subramaniyan V, Chitra V, Fuloria NK, et al. Exploring the contribution of pro-inflammatory cytokines to impaired wound healing in diabetes. Front Immunol. 2023;14:1216321. https://doi.org/10.3389/fimmu.2023.1216321
31. Jiang G, Jiang T, Chen J, Yao H, Mao R, Yang X, et al. Mitochondrial dysfunction and oxidative stress in diabetic wound. J Biochem Mol Toxicol. 2023;37(7):e23407. https://doi.org/10.1002/jbt.23407
32. Sen CK, Roy S, Khanna S. Diabetic peripheral neuropathy associated with foot ulcer: one of a kind. Antioxid Redox Signal. 2023. https://doi.org/10.1089/ars.2022.0093
33. Qazi N, Memon S, Memon F, Goswami P, Sirhandi BR, Goswami B. Antioxidant and hepato-protective effects of ginger in comparison with atorvastatin in hyperlipidemic albino mice. JMMC. 2024;15(1):21–6. https://doi.org/10.62118/jmmc.v15i1.464
34. Shahani SB, Shaikh P, Memon SG, Memon S. Possible prevention of reactive oxygen species induced human trabecular meshwork cell damage by resveratrol and ascorbic acid. Prof Med J. 2019;26(7):1036–41. https://doi.org/10.29309/TPMJ/2019.26.07.3229
35. Memon SG, Shaikh P, Shahani MY, Bano U, Rani S, Memon S. Possible protection against cisplatin induced behavioral changes by dietary antioxidants in adult albino mice. Prof Med J. 2020;27(6):1217–23. https://doi.org/10.29309/TPMJ/2020.27.06.4162
36. An Y, Xu BT, Wan SR, Ma XM, Long Y, Xu Y, et al. The role of oxidative stress in diabetes mellitus-induced vascular endothelial dysfunction. Cardiovasc Diabetol. 2023;22(1):237. https://doi.org/10.1186/s12933-023-01965-7
37. Li X, Zou J, Lin A, Chi J, Hao H, Chen H, et al. Oxidative stress, endothelial dysfunction, and N-acetylcysteine in type 2 diabetes mellitus. Antioxid Redox Signal. 2024;40(16–18):968–989. https://doi.org/10.1089/ars.2023.0524
38. Dou B, Zhu Y, Sun M, Wang L, Tang Y, Tian S, et al. Mechanisms of flavonoids and their derivatives in endothelial dysfunction induced by oxidative stress in diabetes. Molecules. 2024;29(14):3265. https://doi.org/10.3390/molecules29143265
39. Memon SZ, Memon SH, Kazi S, Arain ZI, Memon AG, Hashmi SF. Frequency of hyperuricemia in patients with acute myocardial infarction. Prof Med J. 2020;27(2):284–288. https://doi.org/10.29309/TPMJ/2020.27.02.3500
40. Bang FS, Leeberg V, Ding M, Dreyer CH. The effect of VEGF stimulation in diabetic foot ulcers: a systematic review. Wound Repair Regen. 2024;32(4):384–392. https://doi.org/10.1111/wrr.13171
41. Schönborn M, Łączak P, Pasieka P, Borys S, Płotek A, Maga P. Pro- and anti-angiogenic factors: their relevance in diabetic foot syndrome—a review. Angiology. 2022;73(4):299–311. https://doi.org/10.1177/00033197211042684
42. Wan J, Bao Y, Hou LJ, Li GJ, Du LJ, Ma ZH, et al. lncRNA ANRIL accelerates wound healing in diabetic foot ulcers via modulating HIF1A/VEGFA signaling through interacting with FUS. J Gene Med. 2023;25(2):e3462. https://doi.org/10.1002/jgm.3462
43. Zhao Q, Xu J, Han X, Zhang Z, Qu J, Cheng Z. Growth differentiation factor 10 induces angiogenesis to promote wound healing in rats with diabetic foot ulcers by activating TGF-β1/Smad3 signaling pathway. Front Endocrinol (Lausanne). 2023;13:1013018. https://doi.org/10.3389/fendo.2022.1013018
44. Patel M, Patel V, Shah U, Patel A. Molecular pathology and therapeutics of the diabetic foot ulcer: comprehensive reviews. Arch Physiol Biochem. 2024;130(5):591–598. https://doi.org/10.1080/13813455.2023.2219863
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Nimra Asghar, Zuha Tariq, Minahil Tahir, Fatima Ali, Muhammad Akram, Ejaz Ehsan Khan, Khaleeq Ullah (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





